Abstract
Background: Physicians were directed to prioritize using nonsurgical cancer treatment at the beginning of the COVID-19 pandemic. We sought to quantify the impact of this policy on the modality of first cancer treatment (surgery, chemotherapy, radiotherapy or no treatment).
Methods: In this population-based study using Ontario data from linked administrative databases, we identified adults diagnosed with cancer from January 2016 to November 2020 and their first cancer treatment received within 1 year postdiagnosis. Segmented Poisson regressions were applied to each modality to estimate the change in mean 1-year recipient volume per thousand patients (rate) at the start of the pandemic (the week of Mar. 15, 2020) and change in the weekly trend in rate during the pandemic (Mar. 15, 2020, to Nov. 7, 2020) relative to before the pandemic (Jan. 3, 2016, to Mar. 14, 2020).
Results: We included 321 535 people diagnosed with cancer. During the first week of the COVID-19 pandemic, the mean rate of receiving upfront surgery over the next year declined by 9% (rate ratio 0.91, 95% confidence interval [CI] 0.88–0.95), and chemotherapy and radiotherapy rates rose by 30% (rate ratio 1.30, 95% CI 1.23–1.36) and 13% (rate ratio 1.13, 95% CI 1.07–1.19), respectively. Subsequently, the 1-year rate of upfront surgery increased at 0.4% for each week (rate ratio 1.004, 95% CI 1.002–1.006), and chemotherapy and radiotherapy rates decreased by 0.9% (rate ratio 0.991, 95% CI 0.989–0.994) and 0.4% (rate ratio 0.996, 95% CI 0.994–0.998), respectively, per week. Rates of each modality resumed to prepandemic levels at 24–31 weeks into the pandemic.
Interpretation: An immediate and sustained increase in use of nonsurgical therapy as the first cancer treatment occurred during the first 8 months of the COVID-19 pandemic in Ontario. Further research is needed to understand the consequences.
The COVID-19 pandemic has put cancer treatment systems around the world under immense pressure, forcing redefinitions of care processes to cope with resource shortages and social distancing policies.1 For patients who received a cancer diagnosis during the pandemic, decisions on treatment became more complex, now taking into account risks of SARS-CoV-2 infection, travel restrictions and reduced inpatient capacity, in addition to other patient, disease and system factors. In anticipation of substantial delays in elective cancer surgery, physicians were directed to give surgical priority to a small group of patients and use nonsurgical therapy to a larger extent for others.2,3 Evidence is scarce on the real-world population-level impact of those polices on newly diagnosed cancer. Preliminary data have shown a decreased use of upfront surgery for cancers of the breast4,5 and pancreas6 diagnosed during the pandemic, whereas no difference in first treatment modalities was reported for head and neck,7 liver,8 colorectal9 and digestive system cancers.10 These existing studies are limited by their small samples (most had single-centre designs) and the focus on a single cancer site. Comprehensive data are lacking on this topic for all major cancer types.
In this study, we used data from Ontario, Canada, to contribute to better understanding of the shifts in first cancer treatment modalities (surgery, chemotherapy, radiotherapy or no treatment) used during the pandemic. We aimed to assess the effect of initial pandemic control measures on the first-year management of patients whose cancer diagnosis occurred during the first 8 months of the pandemic. This information is needed for assessing the implementation of the first set of pandemic de-escalation policies to guide policy decisions in the subsequent pandemic waves and future health system emergencies.
Methods
Ontario is Canada’s most populous province, with 14.7 million residents accessing health services under a single-payer health insurance system (Ontario Health Insurance Plan [OHIP]).11 On Jan. 25, 2020, Ontario reported Canada’s first COVID-19 case. On Mar. 15, 2020, all hospitals were directed by the province’s chief medical officer of health to halt nonemergent or elective procedures.12 Around the same time, Ontario Health (Cancer Care Ontario) developed clinical guideline documents to advise hospitals and physicians in the face of reduced surgical capacity; these living documents have been continuously updated to reflect the evolving COVID-19 situation.2,3 Surgical priority was first given to cancer (and cardiology) patients and further to those with an immediate threat to life or limb; with obstructing, bleeding or perforating cancer; or with progressive disease under neoadjuvant systematic therapy (Priority A).2,3 For other patients with cancer (Priority B–C), physicians were advised to consider alternatives to surgery for treatment. Strategic resumption of surgery started in May 2020.13
Study design and population
In this retrospective population-based cohort analysis, the cohort comprised OHIP-eligible adults (≥ 18 yr) diagnosed with cancer between Jan. 3, 2016, and Nov. 7, 2020 (Appendix 1, available at www.cmajopen.ca/content/11/3/E426/suppl/DC1). Only first cancer diagnoses over this period were considered. Each patient was followed for 1 year after date of cancer diagnosis or until date of death. Patients with 2 consecutive cancer diagnoses within 1 year or multiple types of cancer diagnosed on the same day were excluded. We excluded patients with melanoma and skin cancer, as these cancers are frequently treated in the outpatient setting. We also excluded patients with cancers primarily labelled as ophthalmologic and paraneoplastic neurologic syndromes owing to their rarity.
Data sources
Cancer diagnoses were retrieved from the Ontario Cancer Registry, which captures 98% of cancer cases across the province. 14,15 At the time of analysis (August 2022), the Ontario Cancer Registry was updated to November 2021, with nearly complete data until June 2021. Receipt of cancer-directed surgery was determined from the Discharge Abstract Database and Same Day Surgery Database of the Canadian Institute for Health Information and confirmed with the diagnosis records from the Ontario Cancer Registry to ensure the surgical procedure matched with the cancer site and that the procedure was a resection rather than a biopsy.16,17 Radiotherapy and chemotherapy visits were determined using physician billing from the OHIP claims database. Individuals who immigrated to Ontario from January 1985 to May 2017 were identified from the Immigration, Refugees and Citizenship Canada Permanent Residents Database (with data from that period). Rurality was determined from Statistics Canada’s Postal Code Conversion File and defined as living in rural areas or small towns with an urban population of less than 10 000.18 Material deprivation was calculated using the Ontario Marginalization Index.19 These data sets were linked using unique encoded identifiers and analyzed at ICES.
Outcome
For each patient, we looked forward 1 year from date of diagnosis to determine the modality of first cancer treatment: surgery, chemotherapy, radiotherapy or no treatment during the first postdiagnosis year (Appendix 2, available at www.cmajopen.ca/content/11/3/E426/suppl/DC1). For patients receiving hormonal therapy as the first cancer treatment, we also classified them as receiving no treatment since this procedure was not fully captured in the OHIP claims database; this was further assessed in a sensitivity analysis. For patients receiving more than 1 modality of first cancer treatment on the same day, if 1 of those modalities was surgery, we assumed the other nonsurgical modality had been administered before surgery on that day. If chemotherapy and radiotherapy occurred on the same day, we assigned radiotherapy as the first modality.
Statistical analysis
Using Mar. 15, 2020, as the start of the pandemic, we created a prepandemic period (Jan. 3, 2016, to Mar. 14, 2020) and a pandemic period (Mar. 15, 2020, to Nov. 7, 2020).12 We compared characteristics of patients diagnosed in the 2 periods and considered a standardized difference exceeding 0.10 to indicate a significant imbalance.20
For each modality of first cancer treatment, we conducted separate segmented Poisson regression analyses to examine trends in crude rates, defined as the weekly number of recipients (over the next year) per thousand patients diagnosed with cancer (during this week). Three parameter estimates were of interest: the prepandemic weekly trend (slope) in rate, the immediate change in mean rate at the start of the pandemic (relative change in intercept) and further change in slope during the pandemic. 16,21–25 Overdispersion was ruled out (Appendix 3, available at www.cmajopen.ca/content/11/3/E426/suppl/DC1). In a sensitivity analysis, we repeated the regression analysis by excluding patients diagnosed with breast or prostate cancer to assess the impact of not capturing hormonal therapy as the first treatment for some of these patients. Analyses were 2-sided and significance was set at a p value of less than 0.05. Analyses were performed on SAS Enterprise Guide 7.15 (SAS Institute).
Ethics approval
The use of the data in this study is authorized under section 45 of Ontario’s Personal Health Information Protection Act and does not require review by a research ethics board.
Results
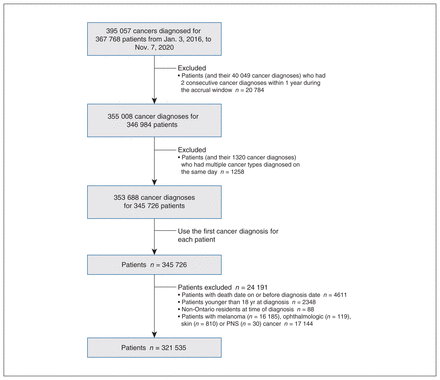
We included 321 535 people with cancer in the study cohort (Figure 1, Table 1). Among them, the vast majority (n = 285 973, 88.9%) were diagnosed in the prepandemic period, and 11.1% (n = 35 562) of patients received a cancer diagnosis during the pandemic. Assuming the OHIP-eligible adult population in Ontario remained at 14.74 million (2018 level) during 2016–2020, this means the annual cancer incidence rate (per 100 000 population) was 461.9 in the prepandemic period and 370.0 in the pandemic period. We did not detect any significant difference of sociodemographic and clinical characteristics between the 2 groups of patients. When we followed all patients for 1 year after diagnosis, those diagnosed during the pandemic were significantly more likely to receive chemotherapy as first treatment than their prepandemic counterparts (20.4% v. 16.7%, standardized difference 0.10).
Flow diagram showing the creation of study cohort. Patients with a diagnosis of melanoma or skin cancer were excluded to ensure a robust capture of cancer-directed surgical procedures performed at a hospital since these cancers are frequently treated in a clinic setting. People with ophthalmologic and PNS cancer were also excluded owing to their small numbers. The Ontario Cancer Registry had cancer incidence data until November 2021 (with nearly complete data available until June 2021) at the time of this analysis (August 2022). Note: PNS = paraneoplastic neurologic syndrome.
Characteristics of patients diagnosed with cancer during prepandemic and pandemic periods (n = 321 535)
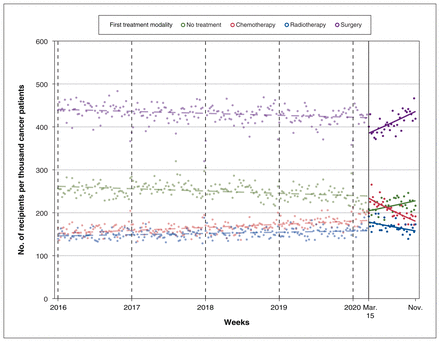
During the first week of the COVID-19 pandemic, the mean rate of receiving upfront surgery over the next year declined immediately by 9% (rate ratio 0.91, 95% confidence interval [CI] 0.88–0.95), and mean rates of chemotherapy and radiotherapy increased by 30% (rate ratio 1.30, 95% CI 1.23–1.36) and 13% (rate ratio 1.13, 95% CI 1.07–1.19), respectively (Table 2). The rate of receiving no treatment in the first postdiagnosis year dropped by 15% (rate ratio 0.85, 95% CI 0.80–0.91) at the start of the pandemic.
Impact of the COVID-19 pandemic on the weekly number of first cancer treatment recipients per thousand people with cancer*
During the pandemic period (Mar. 15 to Nov. 7, 2020), the rate of receiving upfront surgery in the next year increased further by 0.4% (rate ratio 1.004, 95% CI 1.002–1.006) for each week, and during the week of Aug. 23–29, 2020 (the 24th week since the start of the pandemic), the surgical rate had recovered to its prepandemic levels (Figure 2). Upfront chemotherapy and radiotherapy rates decreased (after their initial rise), with a weekly decrement of 0.9% (rate ratio 0.991, 95% CI 0.989–0.994) and 0.4% (rate ratio 0.996, 95% CI 0.994–0.998). For chemotherapy, the rate had fully reduced to prepandemic levels 30 weeks after the start of the COVID-19 pandemic (Oct. 4–10, 2020), and the rate of radiotherapy resumed to prepandemic levels a week thereafter (Oct. 11–17, 2020). The rate of receiving no treatment increased by 0.4% (rate ratio 1.004, 95% CI 1.001–1.006) per week during the pandemic and returned to prepandemic levels on Jan. 10–16, 2021. Excluding patients with breast (n = 49 541) and prostate (n = 39 055) cancer from the cohort did not modify these findings (Appendix 4, available at www.cmajopen.ca/content/11/3/E426/suppl/DC1).
Number of patients by first treatment received within 1 year after diagnosis, per 1000 cancer cases. Trends in the modality of first cancer treatment during prepandemic and pandemic periods in Ontario, Canada. We used dots and lines to denote the observed and predicted weekly recipient volume of first cancer treatment (over the next year) per thousand people diagnosed with cancer (in the current week). The x-axis represents the time of cancer diagnosis. Upfront surgery, chemotherapy and radiotherapy rates within 1 year postdiagnosis have fully recovered to prepandemic levels at the week of Aug. 23–29, 2020, the week of Oct. 4–10, 2020, and the week of Oct. 11–17, 2020, respectively. The rate of receiving no cancer treatment over the next year has resumed during the week of Jan. 10–16, 2021 (not shown).
Interpretation
This population-based cohort study examines the impact of the COVID-19 pandemic on the modalities of first cancer treatment. With the arrival of the pandemic, patients diagnosed with cancer during that week had a 9% lower rate of receiving upfront surgery over the next year, with a corresponding rise in the rate of first receiving nonsurgical therapy, especially chemotherapy. It took 24 weeks for the surgical rate to return to the prepandemic weekly utilization level, and another 6–7 weeks for chemotherapy and radiotherapy rates to fully resume.
The drop in 1-year upfront surgical rate among those diagnosed with cancer during the first week of the COVID-19 pandemic is in line with the provincial mandate to defer elective cancer surgery for patients whose outcomes would not be affected by a delay of 4 weeks (Priority B) to 2 months (Priority C), and is therefore not suprising.3 Indeed, these results show that Ontario physicians made swift and sustained adjustments in treating newly diagnosed cancer, similar to how they had quickly adopted telemedicine when the pandemic started.22,26 We have previously reported a large volume reduction in cancer incidence (34%) and cancer-directed surgery (60%) in Ontario at the beginning of the COVID-19 pandemic;16,17,21 as such, the present analysis additionally shows that patients who were able to get a cancer diagnosis during the first week of the pandemic had a lower chance of receiving upfront surgery over the next year. Although we did not find the profile of patients being diagnosed in the pandemic to differ from the profile of those before, cancer staging was not assessed owing to unavailable data. In light of Ontario’s 2-month suspension of cancer screening programs27 and evidence from elsewhere that more patients presented with advanced-stage, inoperable cancer during the pandemic,28 the resulting shift in the case-mix of incident cancers may contribute to the decrease in rate of upfront surgery. Future studies should quantify the proportion of patients who would have had surgery first but received neoadjuvant therapy instead during the pandemic. These data are required to identify potentially at-risk patients who might experience the negative consequences of surgical delays so that physicians can plan care accordingly to mitigate those repercussions.29
The rate of chemotherapy use as initial treatment increased by 30% for patients with cancer diagnosed during the first week of the pandemic, and for the next 30 weeks (until Oct. 4–10, 2020) chemotherapy utilization remained higher than prepandemic levels. These results contribute to existing evidence on the expanded use of neoadjuvant chemotherapy for patients who would have received surgery upfront.4,30–32 In Ontario, patients initially treated with chemotherapy indeed had an increased receipt of subsequent surgery during the pandemic than before.33 Another possibility is that, owing to stage migration, patients diagnosed during the pandemic were more likely to require palliative chemotherapy or chemotherapy combined with radiotherapy.28 The increased uptake of chemotherapy may also be attributed to the expanded public insurance coverage for hospital-administered cancer drugs in Ontario that was introduced shortly after the start of the COVID-19 pandemic.34 It is important to examine to what extent this temporary increase in neoadjuvant chemotherapy use will affect future oncologic practice, particularly if adopting such practice changes will help physicians and the cancer system to tackle the incoming tsunami of new cancer patients who may be sicker and more challenging to treat. Having an influx of patients starting chemotherapy also has safety implications, as a meta-analysis study found past-month use of chemotherapy to be associated with elevated risk of COVID-19-related death.35 Further study that reports on outcomes among patients receiving chemotherapy during the pandemic is required to guide clinical and drug-funding policies.
We observed a 15% decrease in the mean rate of no treatment over the first postdiagnosis year at the beginning of the pandemic. Owing to the drop in cancer incidence,21 the demand for cancer therapy was lowered, which, coupled with the large-scale cancelation of elective surgeries, may have preserved capacity within the cancer system for nonsurgical care delivery.36 If the pandemic has indeed caused a pronounced migration toward more advanced cancer stages at presentation,28,37 this would translate to more patients requiring immediate treatment. We used a conservative definition for “no treatment” by not counting hormonal therapy, which may result in an underestimation for treatment delivered as bridging therapy before definitive treatment,32 although results of our sensitivity analysis excluding hormone-sensitive cancers suggested this effect to be negligible.
Limitations
Our analyses were not stratified by cancer type or stage, and these data were also unavailable owing to a lag in data acquisition. This study evaluates only the modality of first treatment without evaluating wait times or the subsequent treatment; we addressed these objectives using a time-to-event approach elsewhere.33,38 We assumed patients who received chemotherapy and radiotherapy on the same day (without prior surgery) to always have started radiotherapy first. Although this simplification applies to most patients, some recipients of concurrent chemoradiotherapy were given chemotherapy as a radiosensitizer on the first day of radiotherapy. Future study should examine these nuances of combined modality treatment. As we aimed to assess the initial pandemic control measures, our pandemic cohort only comprised patients diagnosed with cancer during the first 8 months of the COVID-19 pandemic. This short time window did not allow us to reliably assess seasonality.39 Research with more recent data should explicate the effect of subsequent pandemic policies on cancer management.23,25 Future work should also address how these shifts in treatment may have affected outcomes such as survival and recurrence; however, such data will take time to accrue.
Conclusion
Compared with the prepandemic period, patients with cancer diagnosed during the first week of the COVID-19 pandemic in Ontario had a 9% lower rate of receiving treatment with upfront surgery over the next year, while having a 30% higher rate of receiving chemotherapy and a 13% higher rate of receiving radiotherapy first. Meanwhile, the mean rate of no treatment over the first postdiagnosis year decreased by 15%, possibly owing to both decrease in cancer incidence and a shifted case-mix of new patients. Twenty-four weeks into the pandemic, the surgery rate had returned to prepandemic levels, and it took another 6–7 weeks for chemotherapy and radiotherapy to return to prepandemic levels. The findings of this study suggest a sustained deviation from the standard of care for many patients, with an unknown impact on recurrence, quality of life and survival. Health care systems should work toward preserving resources to manage newly diagnosed cancer according to standard of care.
Footnotes
Competing interests: Jonathan Irish is a member of the Canadian Cancer Society and the Ontario Institute for Cancer Research. Natalie Coburn is the clinical lead for patient-reported outcomes and symptom management at Ontario Health (Cancer Care Ontario). Julie Hallet reports receiving speaking honoraria from Ipsen Biopharmaceuticals, Advanced Accelerator Applications, Medtronic and Bristol-Myers Squibb. Monika Krzyzanowska reports receiving consulting fees from Lilly and Ipsen and attending the advisory boards of Ipsen and Bayer. Alexander Louie reports receiving speaking honoraria and being on the advisory board of AstraZeneca. Nicole Look Hong reports receiving consulting fees from MOLLI Surgical. Daniel McIsaac is on the advisory board of the PRICE 2 Trial and is a member of the American Society of Anesthesiology Committee on Geriatric Anesthesia. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Antoine Eskander and Rinku Sutradhar had full access to the study data and take responsibility for the integrity of the data and the accuracy of the data analysis. Qing Li was involved in the data analysis. Rui Fu led the drafting of the manuscript. All authors contributed to the conception of the study, interpretation of the data and review of the manuscript. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This work was supported by a Sunnybrook Research Institute and Sunnybrook Foundation COVID-19 Response Grant and a Canadian Institutes of Health Research Operating Grant no. 179892. This work was also supported by the Ontario Health Data Platform (OHDP), a Province of Ontario initiative to support Ontario’s ongoing response to COVID-19 and its related impacts. The funders were not involved in any part of the study, including design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review or approval of the manuscript; and decision to submit the manuscript for publication.
Data sharing: The data set from this study is held securely in coded form at ICES. While legal data-sharing agreements between ICES and data providers (e.g., health care organizations and government) prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS (email: das{at}ices.on.ca). The full data set creation plan and underlying analytic code are available from the authors on request, understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). This document used data adapted from the Statistics Canada Postal CodeOM Conversion File version 7B (November 2018), which is based on data licensed from Canada Post Corporation and/or data adapted from the Ontario MOH Postal Code Conversion File, which contains data copied under licence from Canada Post Corporation and Statistics Canada. This does not constitute an endorsement by Statistics Canada of this product. Parts of this material are based on data and/or information compiled and provided by the Ontario MOH, Ontario Health, the Canadian Institute for Health Information and Immigration, Refugees and Citizenship Canada current to May 31, 2017. Parts of this report are based on Ontario Registrar General (ORG) information on deaths, the original source of which is ServiceOntario. The views expressed therein are those of the author and do not necessarily reflect those of ORG or the Ministry of Public and Business Service Delivery. The authors thank the Toronto Community Health Profiles Partnership for providing access to the Ontario Marginalization Index. The analyses, conclusions and opinions expressed therein are those of the authors and do not necessarily reflect those of the funding or data sources; no endorsement is intended or should be inferred. No endorsement by the OHDP, its partners, or the Province of Ontario is intended or should be inferred.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/3/E426/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors