Abstract
Background: Given recent discrepant results from randomized controlled trials (RCTs), we examined the totality of RCT evidence assessing the association between dipeptidyl peptidase-4 (DPP-4) inhibitors and heart failure.
Methods: MEDLINE, Embase and ClinicalTrials.gov were searched without language restrictions to August 2016 for RCTs comparing DPP-4 inhibitors to placebo or no therapy for a period of 24 weeks or more. We included all heart failure outcomes when listed either as a serious adverse event or adverse event. Pooled analyses used random-effects.
Results: We identified 100 RCTs (n = 79 867) - 3 large cardiovascular-safety RCTs (SAVOR-TIMI 53[saxagliptin]/n = 16 492, EXAMINE[alogliptin]/n = 5380, and TECOS[sitagliptin]/n = 14 735), and 97 smaller RCTs with a primary outcome that was usually change in glycated hemoglobin. Virtually all RCTs were high-quality, multicentre, placebo-controlled trials. A total of 96% (1192/1244) of heart failure events were prespecified, blindly adjudicated and required hospital admission. Pooled results suggested a 13% increase in heart failure (relative risk [RR] 1.13, 95% confidence interval [CI] 1.01-1.26, I2 = 0%; 32 RCTs, n = 54 640, 1244 events). When including only the 3 large RCTs, the increase was similar, but not significant (RR 1.14, 95% CI 0.97-1.32; 3 RCTs, n = 36 543, 1169 adjudicated events; number needed to harm 246) owing to heterogeneity (I2 = 42%), which lead to wider CIs, because SAVOR-TIMI 53 showed increased heart failure (RR 1.26, 95% CI 1.06-1.49) and TECOS showed no effect (RR 1.00, 95% CI 0.83-1.19).
Interpretation: Despite pooled data from 79 867 patients, whether DPP-4 inhibitors increase heart failure overall or exhibit within-class differences remains unresolved. Our results highlight the importance of ongoing trials that are comparing DPP-4 inhibitors to placebo, although no large cardiovascular-safety RCTs are comparing different DPP-4 inhibitors to each other; consequently, these will address the overall but not class-difference question.
Dipeptidyl peptidase-4 (DPP-4) inhibitors are integral in the management of diabetes because of their effective glucose lowering with low risk for hypoglycemia or weight gain.1 Because heart failure remains a common complication of diabetes and is associated with poor long-term prognosis,2,3 understanding the potential effects of antihyperglycemic agents on risk for heart failure is of critical and immediate importance. The first large DPP-4 inhibitor versus placebo randomized controlled trial (RCT) Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus - Thrombolysis in Myocardial Infarction 53 (SAVOR-TIMI 53) (n = 16 492 patients with a history of, or at risk for, cardiovascular events) unexpectedly found a significantly higher rate of heart failure requiring admission to hospital.4,5 The second was the Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care (EXAMINE) (n = 5380 patients post-acute coronary syndrome), which found a numerical but nonsignificantly higher rate of heart failure requiring hospital admission.6,7 In contrast, Trial to Evaluate Cardiovascular Outcomes after Treatment with Sitagliptin (TECOS) (n = 14 735 patients with cardiovascular disease and longer follow-up [median 3.0 yr v. 1.5 and 2.1 yr for EXAMINE and SAVOR-TIMI 53]) found almost identical rates of hospital admission for heart failure in the sitagliptin and placebo groups.8
The potential safety issue that arose from SAVOR-TIMI 53 and EXAMINE led to the Food and Drug Administration's (FDA) recommendation9 to consider discontinuing saxagliptin and alogliptin for patients if heart failure develops. Given the apparent discrepant results from TECOS,3,10,11 we felt it was important to inform clinicians who are concerned about the potential increased heart failure signal by providing them with the totality of the available RCT evidence in the field. In addition, the publication of the Empagliflozin, Cardiovascular Outcomes, and Mortality in type 2 Diabetes [EMPA-REG OUTCOMES] trial,12 which shows that hopspital admission for heart failure was significantly reduced with the use of an oral antihyperglycemic agent of a different class, empagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor, has increased the importance of quantifying the risk of increased heart failure for DPP-4 inhibitors. The 2 specific questions addressed by this systematic review and meta-analysis are whether DPP-4 inhibitors, as a class, compared with placebo or no therapy, increases heart failure in patients with type 2 diabetes, and whether there are significant within-class differences.
Methods
Data sources and study selection
We systematically searched MEDLINE and Embase (inception to August 2016) and ClinicalTrials.gov in duplicate for RCTs that compared treatment with any DPP-4 inhibitor with either placebo or no therapy (active comparator RCTs were excluded) and that enrolled adult patients with type 2 diabetes for at least 24 weeks. For multiple treatment group RCTs, we included only randomized groups in which treatments differed by DPP-4 inhibitor treatment. Groups with different DPP-4 inhibitor doses were combined within the same trial. Trials in which placebo groups were subsequently switched to open-label active therapy were only included if this switch occurred after 24 weeks of therapy.
Data extraction and risk of bias assessment
For each RCT, baseline patient characteristics, intervention, outcome definitions and events were collected in duplicate (discrepancies resolved by consensus). Risk of bias (patient, caregiver and outcome assessor blinding; allocation concealment; intention-to-treat analysis; early stopping for benefit;13 loss to follow-up) were also assessed in duplicate.14
Data analysis
In the primary analysis, we included all heart failure outcomes when listed either as a serious adverse event or adverse event. In 2 separate secondary analyses, we included only RCTs in which (1) cardiovascular outcomes were the primary outcome, and (2) hospital admission for heart failure was an adjudicated primary or secondary outcome. Additional data analysis details, including sensitivity analysis, are provided in the online appendix (Appendix 1, available at www.cmajopen.ca/content/5/1/E152/suppl/DC1). We did not register or publish a review protocol.
Results
Search results
We identified 121 RCTs in which treatment between randomized groups differed only by DPP-4 inhibitor treatment. Of these, 11 RCTs listed only on ClinicalTrials.gov provided no results (NCT00683735, NCT01356381, NCT01582230, NCT01697592, NCT01704261, NCT01792518, NCT01890122, NCT01990469, NCT02015299, NCT02099110, NCT02104804) and 10 RCT publications did not provide heart failure data,15-24 leaving 100 RCTs that reported the number of patients with heart failure (Appendix 1, Figure 1), which enrolled 79 867 patients into groups that differed only in DPP-4 inhibitor therapy.4-8,25-47,49-88,90-95,97-139
Search strategy and trial flow.
RCT patient characteristics
Only 3 RCTs, SAVOR-TIMI 53, EXAMINE and TECOS, had cardiovascular outcomes as the primary outcome; they enrolled 46% (36 543/79 867) of all patients in the included RCTs (Table 1, Table 2). Enrolled patients in these 3 RCTs had a mean age of 61-66 years, two-thirds of participants were men and about 70% were white. Median body mass index (BMI) was about 30 kg/m2. The patients had type 2 diabetes for median durations that ranged from 7-10 years, with a mean baseline glycated hemoglobin (A1C) of 8.0% (SAVOR-TIMI 53, EXAMINE) or 7.2% (TECOS), and a mean of 23%-41% participants taking insulin in addition to oral antihyperglycemic agents. Many patients had other cardiovascular risk factors, including hypertension and dyslipidemia, and most had a previous myocardial infarction or revascularization, although only a few had a previous stroke (Table 2). Most patients were receiving acetylsalicylic acid (ASA) with or without another antiplatelet therapy, as well as a statin, angiotensin-converting enzyme inhibitors or angiotensin receptor antagonists, and β-blockers or other antihypertensive medications. The proportion of patients with pre-existing heart failure ranged from 13%-28% among the included larger RCTs.
The primary outcome for most of the smaller RCTs (n = 89/97) was glycemic control. One small trial, presented only in abstract form,113 enrolled patients with New York Heart Association class I-III heart failure and measured change in left ventricular ejection fraction as its primary outcome. Enrolled patients in these smaller trials had variable characteristics (Table 1). Mean age was typically in the mid-50s, with more than 50% men, and more than 50% of white race, and median BMI was about 30 kg/m2. The mean duration of type 2 diabetes ranged from 0 to 17 years, with most mean baseline A1C levels between 8.0% and 8.5%. No patients were using insulin therapy in most of the smaller RCTs. Few data were provided on the prevalence of other cardiovascular risk factors or cardiovascular medication use in the smaller RCTs.
Risk of bias assessment
Included RCTs generally had low risk of bias. The 3 large RCTs and all trials with events were blinded using placebos with concealed allocation, intention-to-treat analysis, no stopping early for benefit and had low numbers (typically < 1%-5%) of randomized patients with missing heart failure outcome data (Table 3). Only 7 of the smaller trials, each with 0 events, were not blinded or had unclear allocation concealment,34,80,101,106,108,129,133, and only 4 had more than 5% of randomized patients lost to follow-up.34,88,125,129 The 3 larger trials, in addition to the small trial that enrolled patients with class I-III heart failure,113 defined heart failure that required admission to hospital as a prespecified secondary outcome that was adjudicated by outcome assessors blinded to treatment allocation. The 3 larger trials used virtually identical definitions of heart failure (Table 2). Virtually all RCTs were funded by pharmaceutical companies.
Quantitative data synthesis
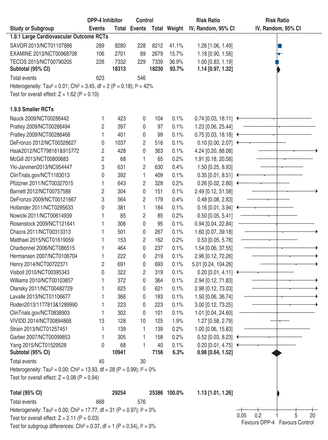
All heart failure outcomes were listed as serious adverse events. Pooling heart failure data from the 3 larger RCTs and the 29 smaller RCTs with at least 1 patient with heart failure suggested a 13% increased risk of heart failure with DPP-4 inhibitors, which achieved significance (pooled relative risk [RR] 1.13, 95% confidence interval [CI] 1.01-1.26, p = 0.03; 32 RCTs, 54 640 patients, 1244 events) with no significant heterogeneity (I2 = 0%) (Figure 2). A total of 94% (1169/1244) of the events came from the 3 larger RCTs, and 23 of the remaining 75 (31%) events reported in the 29 smaller RCTs occurred in the trial that enrolled patients with class I-III heart failure. Thus, 1192/1244 (96%) of heart failure outcomes occurred in RCTs that prespecified that these required admission to hospital and were subject to blinded adjudication. Including the results from the 68 smaller RCTs (n = 25 227) with no patients with heart failure and thus an RR of 1.00 (or no effect), as the preplanned sensitivity analysis, did not change the pooled result (pooled RR 1.12, 95% CI 1.01-1.25, p = 0.03, I2 = 0%; 100 RCTs, 79 867 patients, 1244 events).
Forest plot for heart failure, large versus small trials. Individual and pooled risk ratios (RRs) with 95% confidence intervals (CIs) for randomized controlled trials (RCTs) with a primary outcome that included cardiovascular outcomes and reported the number of patients in each treatment group that were admitted to hospital for heart failure as an adjudicated primary or secondary outcome, as well as smaller RCTs reporting at least 1 patient with heart failure for which outcomes were not necessarily adjudicated and patients not necessarily admitted to hospital. The pooled RRs with 95% CIs were calculated using random-effects models. Weight refers to the contribution of each study to the overall pooled estimate of treatment effect. Each square and horizontal line denotes the point estimate and 95% CI for each trial's RR. The diamonds signify the pooled RR; the diamond's centre denotes the point estimate and width denotes the 95% CI.
Pooling data from only the 3 large RCTs with cardiovascular primary outcomes and blinded outcome adjudication, as the preplanned secondary analysis, resulted in a similar effect size; however, this did not achieve significance (pooled RR 1.14, 95% CI 0.97-1.32, p = 0.10; 3 RCTs, 36 543 patients, 1169 [adjudicated] events), partly because of heterogeneity (I2 = 42%) that resulted in wider CIs (the pooled result would be significant if fixed-effects meta-analysis, which ignores heterogeneity, were used: pooled RR 1.14, 95% CI 1.01-1.27, p = 0.03). Adding the results of the smaller trial that enrolled patients with class I-III New York Heart Association heart failure and defined heart failure that required hospital admission as a prespecified secondary outcome that was adjudicated by outcome assessors blinded to treatment allocation, gives a pooled result that just achieves statistical significance (pooled RR 1.139, 95% CI 1.002-1.293, p = 0.046; 4 RCTs, 36 796 patients, 1192 [adjudicated] events). There was no difference in the pooled result of the 3 larger RCTs with cardiovascular primary outcomes and the pooled result of the smaller RCTs (interaction p = 0.54) (Figure 2).
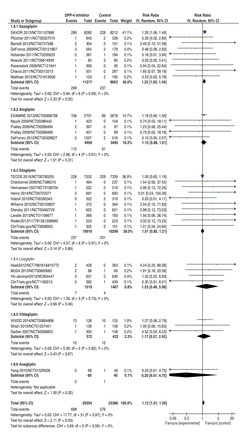
Differences between pooled RR for individual DPP-4 inhibitors (Figure 3) did not achieve significance. The most extreme difference was between saxagliptin, dominated by the results of SAVOR-TIMI 53, which suggested a significantly increased risk of heart failure requiring hospital admission (pooled RR 1.22, 95% CI 1.03-1.44, p = 0.02, I2 = 0%; 9 RCTs, 20 880 patients, 536 events), and sitagliptin, dominated by the results of TECOS, which suggested no difference in risk (pooled RR 1.01, 95% CI 0.85-1.21, p = 0.89, I2 = 0%; 10 RCTs, 21 218 patients, 468 events). However, even this difference in pooled RR between saxagliptin and sitagliptin did not achieve significance (interaction p = 0.13 [interaction p = 0.07 comparing RR for only SAVOR-TIMI 53 v. RR for only TECOS]).
Forest plot for heart failure by DPP-4 inhibitor. Individual and pooled risk ratios (RRs) with 95% confidence intervals (CIs) for larger and smaller randomized controlled trials (RCTs) by DPP-4 inhibitor. Interaction p values comparing RRs between pairs of subgroups of RCTs using different DPP-4 inhibitors were all nonsignificant. For the most extreme difference between saxagliptin RCTs and sitagliptin RCTs, interaction p = 0.13 (interaction p = 0.07 comparing RR for only SAVOR-TIMI 53 v. TECOS). The pooled RRs with 95% CI were calculated using random-effects models. Interaction p values were calculated using Z tests. Weight refers to the contribution of each study to the overall pooled estimate of treatment effect. Each square and horizontal line denotes the point estimate and 95% CI for each trial's RR. The diamonds signify the pooled RR; the diamond's centre denotes the point estimate and width denotes the 95% CI.
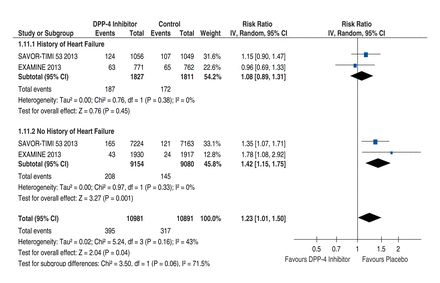
In post hoc analysis, only SAVOR TIMI-53 and EXAMINE provided data for patients with and without a history of heart failure. Rates of heart failure that required hospital admission were considerably higher among patients with (359/3638 [9.9%]) than without (353/3638 [1.9%]) a history of heart failure, but the increase was concentrated in patients without (RR 1.42, 95%CI 1.15-1.74) rather than with (RR 1.08, 95%CI 0.89-1.31) a history of heart failure (interaction p = 0.06) (Figure 4). However, cautious interpretation is needed given the limited data.
Forest plot for heart failure requiring hospital admission by previous history of heart failure. Individual and pooled risk ratios (RRs) with 95% confidence intervals (CIs) for the outcome of heart failure requiring hospital admission in subgroups of patients with versus without previous heart failure in SAVOR-TIMI 53 and EXAMINE, the only RCTs that provided these data. Average rates of heart failure requiring hospital admission were 9.9% ([124+ 107 + 63 + 65]/[1056 + 1049 + 771 + 762] = 359/3638) in patients with versus 1.9% ([165 + 121 + 43 + 24]/[7224 + 7163 + 1930 + 1917] = 353/18 234) in patients without a prior history of heart failure. In this analysis, the heterogeneity in the overall analysis (I2 = 43%) is reduced (I2 = 0%) within each subgroup. The pooled RRs with 95% CIs were calculated using random-effects models. Interaction p values were calculated using Z tests. Weight refers to the contribution of each study to the overall pooled estimate of treatment effect. Each square and horizontal line denotes the point estimate and 95% CI for each trial's RR. The diamonds signify the pooled RR; the diamond's centre denotes the point estimate and width denotes the 95% CI.
Interpretation
Pooled data from all RCTs (n = 79 867) in which intervention and control patients differed only by DPP-4 inhibitor therapy suggest that DPP-4 inhibitors increase the risk of heart failure requiring hospitalization by 13% (1%-26%, p = 0.03). This increase is significant if data from both large and small high-quality RCTs are included (primary analysis), or if data from the 3 large RCTs4-8 and 1 smaller RCT113 that defined heart failure requiring hospital admission as a prespecified secondary outcome adjudicated by outcome assessors blinded to treatment allocation are included (secondary analysis). If, however, data from only the 3 large RCTs with primary cardiovascular outcomes are included,4-8 the magnitude remains numerically similar, but is no longer significant, largely owing to heterogeneity between the risk of heart failure with saxagliptin and sitagliptin. Unfortunately, the current data do not have sufficient statistical power to definitively answer either the question of whether DPP-4 inhibitors as a class increase heart failure given pooled treatment effect p values ranging from 0.03 to 0.10, depending on whether or not the smaller RCT data are included, or whether DPP-4 inhibitors show significant within-class differences (in which case pooling would not be appropriate) given interaction p values ranging from 0.07-0.13 that compare results between the 2 medications with the most extreme safety (sitagliptin) or harm (saxagliptin) results. Results from the 2 ongoing DPP-4 inhibitor versus placebo cardiovascular safety RCTs will be important because they could have an impact on the pooled risk estimates for heart failure among the cardiovascular safety RCTs (see Appendix 1), which emphasizes the importance of ongoing trials to resolve the question of whether DPP-4 inhibitors as a class increase heart failure. Because there are only single large cardiovascular safety RCTs for each specific DPP-4 inhibitor and few head-to-head comparisons (our search identified 11 small short-term RCTs23,109,140-148 directly comparing agents but none reported any heart failure events), the ongoing cardiovascular safety trials will be less helpful in identifying differential effects among DPP-4 inhibitors.
In comparison to previous systematic reviews,149-154 ours focuses on RCTs in which randomized groups differed by DPP-4 inhibitor treatment to avoid the confounding effect of other medications, some of which are known to independently increase or decrease the risk of heart failure,12,155 and is unique in its comparison of differences in heart failure outcomes between different agents (Table 4).
Limitations
Our meta-analysis has limitations. It included relatively small trials with variable inclusion criteria, short follow-up times (although we specified minimum of 24 weeks, which is longer than the 12 weeks follow-up used in some other meta-analyses150,151,153) and non-adjudicated outcomes. However, 96% of the heart failure outcomes were blindly adjudicated, and pooled results are dominated by the large cardiovascular safety trials with adjudicated outcomes: point estimates are similar regardless of whether the data from the smaller trials are included (1.14 v. 1.13), although inclusion of the additional data from the smaller RCTs narrows the confidence intervals, resulting in significance being achieved. This is in contrast to a highly cited rosiglitazone meta-analysis,156 where smaller RCTs with non-adjudicated outcomes drove overall results and the effect on both myocardial infarction and cardiovascular death changed depending on how the analysis was conducted.157 Nevertheless, the absolute increase in risk during follow-up, even limiting the analysis only to the 3 cardiovascular safety RCTs, is small at about 0.4% (623/18 313 to 546/18 230) (Figure 2), corresponding to a number needed to harm of (1/0.004) 246 (median follow-up 2.4 year). Studies involving patients with previous heart failure and longer follow-up data may uncover higher risks, and are needed to explore longer-term safety of these lifelong therapies. Although we limited our analysis to placebo-controlled trials, in trials targeting A1C, patients who received placebo would likely have received more non-DPP-4 inhibitor medications.
Conclusion
In summary, our updated systematic review includes more RCTs than others and is the only post-TECOS meta-analysis to show statistically higher, albeit small, overall heart failure risk, but only if data from all placebo-controlled RCTs are included. However, despite pooled data from 79 867 patients, whether DPP-4 inhibitors increase heart failure overall, or exhibit within-class differences (which would make pooling between agents inappropriate), remains unresolved, which highlights the importance of ongoing trials that will address the overall but not class difference question. Nevertheless, given the current data, it seems prudent to follow the FDA's Drug Safety Communication9 and be cautious about prescribing saxagliptin and alogliptin in patients with established heart failure or at high risk of developing heart failure (previous heart failure, low estimated glomerular filtration rate or elevated N-terminal pro b-type natriuretic peptide), and consider discontinuing these medications in any patient who develops heart failure.
Supplemental information
For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/5/1/E152/suppl/DC1
Footnotes
Competing interests: Subodh Verma has received speaker honoraria and/or grants from Amgen, AstraZeneca, Merck, Novartis, Sanofi, and Valeant. Ronald Goldenberg has received research support from AstraZeneca, Böehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck, Novartis, Novo Nordisk, Takeda; has served on advisory panels for AstraZeneca, Böehringer Ingelheim, Eli Lilly, Merck, Novo Nordisk, and Takeda; has participated in speaker bureaus for AstraZeneca, Böehringer Ingelheim, Eli Lilly, Merck, and Novo Nordisk and Servier; and has served as a consultant for AstraZeneca, Böehringer Ingelheim, Eli Lilly, Merck, Novo Nordisk and Takeda. Deepak Bhatt discloses the following relationships - Advisory Board: Cardax, Elsevier Practice Update Cardiology, Medscape Cardiology, Regado Biosciences; Board of Directors: Boston VA Research Institute, Society of Cardiovascular Patient Care; Chair: American Heart Association Quality Oversight Committee; Data Monitoring Committees: Duke Clinical Research Institute, Harvard Clinical Research Institute, Mayo Clinic, Population Health Research Institute; Honoraria: American College of Cardiology (Senior Associate Editor, Clinical Trials and News, ACC.org), Belvoir Publications (Editor-in-Chief, Harvard Heart Letter), Duke Clinical Research Institute (clinical trial steering committees), Harvard Clinical Research Institute (clinical trial steering committee), HMP Communications (Editor-in-Chief, Journal of Invasive Cardiology), Journal of the American College of Cardiology (Guest Editor; Associate Editor), Population Health Research Institute (clinical trial steering committee), Slack Publications (Chief Medical Editor, Cardiology Today's Intervention), Society of Cardiovascular Patient Care (Secretary/Treasurer), WebMD (CME steering committees); Other: Clinical Cardiology (Deputy Editor), NCDR-ACTION Registry Steering Committee (Vice-Chair), VA CART Research and Publications Committee (Chair); Research Funding: Amarin, AstraZeneca (including for serving as the co-PI of SAVOR-TIMI 53), Bristol-Myers Squibb, Eisai, Ethicon, Forest Laboratories, Ischemix, Medtronic, Pfizer, Roche, Sanofi Aventis, The Medicines Company; Royalties: Elsevier (Editor, Cardiovascular Intervention: A Companion to Braunwald's Heart Disease); Site Co-Investigator: Biotronik, Boston Scientific, St. Jude Medical; Trustee: American College of Cardiology; Unfunded Research: FlowCo, PLx Pharma, Takeda. Kim Connelly has received support from Astra Zeneca/Bristol Myer Squibb, consultancy fees from Servier, Merck, Janssen and Boehringer Ingelheim, travel support from Bristol Myer Squibb and holds a patent with Boehringer Ingelheim for linagliptin and heart failure with preserved ejection fraction. Lawrence Leiter has received research funding from, has provided CME on behalf of, and/or has acted as an advisor to AstraZeneca, Böehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Janssen, Merck, NovoNordisk, Pfizer, Sanofi, and Servier. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.Competing interests: See end of article.
Contributors: Subodh Verma, Ronald Goldenberg and Jan Friedrich contributed to the conception, design, acquisition, analysis and interpretation of data, drafted and revised the article. Deepak Bhatt, Michael Farkouh, Kim Connelly and Lawrence Leiter contributed to data interpretation and revised the article critically for important intellectual content. Adrian Quan and Hwee Teoh contributed to the acquisition of data, drafted and revised the article. All of the authors gave final approval to the version to be published and agree to act as guarantors of the work.
Funding: Subodh Verma is the Canada Research Chair (CRC) in Atherosclerosis. Kim Connelly is supported by a New Investigator award from the CIHR. Jan Friedrich is supported by a Clinician-Scientist Award from the Canadian Institutes of Health Research (CIHR). The CRC program and CIHR had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
References
- Copyright 2017, Joule Inc. or its licensors