Abstract
Background: Deaths from respiratory tract infections (RTIs) in children are preventable through timely access to public health and medical interventions. We aimed to assess whether socioeconomic disparities in mortality related to pediatric RTI persisted after accounting for health status at birth.
Methods: We compared the prevalence of and risk factors for RTI-related death in singletons aged 28 days to 4 years across Ontario (Canada), Scotland and England (jurisdictions with universal health care) using linked administrative data for 2003–2013. We estimated rates of RTI-related mortality for children living in deprived areas and those born to teenage girls; we estimated both crude rates and those adjusted for health status at birth.
Results: A total of 1 299 240 (Ontario), 547 556 (Scotland) and 3 910 401 (England) children were included in the study. Across all jurisdictions, children born in the most deprived areas experienced the highest rates of RTI-related mortality. After adjustment for high-risk chronic conditions and prematurity, we observed differences in mortality according to area-level deprivation in Ontario and England but not in Scotland. In Ontario, teenage motherhood was also an independent risk factor for RTI-related mortality.
Interpretation: Socioeconomic disparities played a substantial role in child mortality related to RTI in all 3 jurisdictions. Context-specific investigations around the mechanisms of this increased risk and development of programs to address socioeconomic disparities are needed.
There have been great strides in reducing childhood mortality over the past century. Globally, the mortality rate among children less than 5 years of age declined by more than 50% during the period of the United Nations Millennium Development Goals, decreasing from 90 to 43 deaths per 1000 live births between 1990 and 2015.1 However, there are still improvements to make, in both developing and developed nations.2,3 Respiratory tract infections (RTIs), such as pneumonia and bronchiolitis, are avoidable causes of mortality and are the third most common cause of death worldwide in children less than 5 years.4–6
It is well known that children born preterm or with certain congenital anomalies or other chronic conditions are at increased risk for both all-cause mortality and RTI-related mortality.7–9 However, deaths from RTIs are considered to be readily preventable through timely access to appropriate public health or medical interventions, including vaccination, passive immunization through palivizumab administration and antibiotic treatment.4,10 In this study, we aimed to assess whether socioeconomic disparities in mortality related to pediatric RTIs in jurisdictions with universal access to physician and hospital care persisted after health status at birth was accounted for.
Methods
We sought to compare the rates of RTI-related death using nationally representative birth cohorts from 3 jurisdictions — Ontario (Canada’s most populous province), Scotland and England — to determine the contribution of socioeconomic risk factors to RTI-related deaths among children less than 5 years of age, taking clinical risk factors at birth into account. These jurisdictions, all generally comparable, high-income, English-speaking regions with publicly funded universal health care systems, have implemented prevention programs for bacterial and viral pathogens causing RTIs (e.g., pertussis, Hemophilus influenzae type B, influenza and pneumococcal) (Appendix 1, Supplemental Table S1, available at www.cmajopen.ca/content/8/2/E273/suppl/DC1).
Study population
We identified outcomes from the age of 28 days until the day before the fifth birthday via linkage between birth registration, hospital admission and mortality registration data sets to create 3 nationally representative birth cohorts. Singleton children born to resident women between Dec. 4, 2002, and Dec. 4, 2012 (i.e., reaching 28 days of age by Jan. 1) were included. We identified outcomes until the earliest of death, moving out of province, the child’s fifth birthday or Dec. 31, 2013 (i.e., end of follow-up). We excluded multiple births since data for these children are prone to linkage error.11 We excluded neonatal deaths (i.e., occurring before 28 d) to reduce bias related to differences in neonatal death certificates, and classification of stillbirths and early neonatal deaths between jurisdictions.12 We excluded babies with a birth weight less than 500 g or born before 24 weeks’ gestation to avoid including stillbirths and to reduce possible bias based on different clinical approaches to resuscitating infants of extremely low birth weight across the 3 jurisdictions.
Data sources
We created the Ontario birth cohort using the MOMBABY database, a linked database of maternal and newborn records covering all hospital births in the province since 1988. MOMBABY is a derived birth registry that has been linked to other health care and death records (linkage rates of mothers and babies exceeded 98% and rate of linkage to death records was 96.2% for the study period)13 by means of a unique coded identifier14 and analyzed at ICES.
We identified the Scottish birth cohort from linked birth registration, maternity records (Scottish Morbidity Record-02) and Scottish Birth Records (which cover neonatal diagnoses), with follow-up through hospital admission (Scottish Morbidity Record-01) and death records. The Scottish databases were linked with the use of the Community Health Index number, a unique personal identifier used in all interactions within the Scottish National Health Service; linkage rates exceeded 99.8% during the study period.15
We derived the birth cohort for England via linkage between delivery and birth records in the Hospital Episode Statistics Admitted Patient Care database, which covers 97% of all births in England.16–18 Birth and hospital admission records and mortality records were linked longitudinally by the data provider, National Health Service Digital, using a deterministic algorithm based on a unique personal identifier. 16 Further details about the linked data sets can be found in Appendix 1, Supplemental Table S2.
Outcomes
We defined a death related to RTI as any noninjury death where an RTI was listed as any cause of death on the death certificate or as any diagnosis on a hospital record with a date of discharge up to 30 days before the date of death. We identified RTIs using International Statistical Classification of Diseases and Related Health Problems, 10th Revision codes A37 (pertussis), A15–A19 (tuberculosis), B97.4 (respiratory syncytial virus) and J00–J22 (acute upper and lower respiratory infections). We defined injurious deaths as deaths where the underlying cause was coded as an external cause, i.e., diagnostic codes V00–Y99. Owing to variabilities in the reporting and coding of confirmed respiratory pathogens, we did not further assess outcomes according to specific respiratory pathogens.
Risk factors
We examined risk of RTI-related death according to age, sex, birth characteristics, presence of high-risk chronic conditions and socioeconomic status, as operationalized through area-level deprivation quintiles and maternal age. We selected these factors given their known association with health outcomes and common availability across all 3 jurisdictions.
All analyses were age-stratified, i.e., 28–364 days and 1–4 years, as there are large differences in the magnitude and drivers of mortality risks in these 2 age groups.19 We derived birth weight from maternity records (Ontario and Scotland) or hospital birth or delivery episodes (England) and subsequently coded it into a 6-level categoric variable: less than 1500 g, 1500–2499 g, 2500–2999 g, 3000–3499 g, 3500–3999 g and 4000 g or more. We coded gestational age into a 5-level categoric variable: early preterm (< 34 wk), late preterm (34–36 wk), early term (37–38 wk), term (39–40 wk) and late term (≥ 41 wk).
Ontario’s criteria for prophylaxis of respiratory syncytial virus prophylaxis are based on recommendations from leading organizations such as the American Academy of Pediatrics20 and the Canadian Paediatric Society.21 We used clinical judgment and these criteria to derive a list of diagnostic codes (Appendix 1, Supplemental Table S3) indicating chronic conditions associated with an increased risk of severe symptoms of RTIs in children, following an existing categorization of pediatric chronic conditions.22,23 To identify these high-risk conditions, we searched any cause of death code recorded on the child’s death certificate or an associated hospital admission (within 30 d of death) where the discharge date was before the child’s first birthday (age 28–364 d) or before the fifth birthday (age 1–4 yr).
Exposure of interest
Socioeconomic status was the main exposure of interest. We defined it using maternal age and small-area–level deprivation. Young maternal age is associated with material disadvantage in all 3 jurisdictions24 and was therefore chosen as an indicator of individual-level deprivation. We coded maternal age as follows: less than 19, 19–24, 25–29, 30–34, 35–39, and 40 or more.
For Ontario, we used the 2006 Ontario Marginalization Index to determine material deprivation level, which combines 6 indicators of material deprivation: lone-parent families; people aged 25 years or more without any postsecondary training; income from social benefits programs; unemployment rates among those aged 15 years or more; people below the poverty line; and number of houses needing a major repair.25 The Ontario Marginalization Index is applied at Canada’s smallest census area, the dissemination area (population 400–700).25
For Scotland, we defined area-level deprivation using the Carstairs index, which combines 4 indicators of material deprivation: social class, household overcrowding, car ownership and male unemployment, as measured by the 2001 and 2011 censuses.26 The Carstairs index applies at the level of postcode sector (average population 5000).26
For England, we defined area-level deprivation using the Index of Multiple Deprivation, which is based on 7 distinct domains: employment; income; health deprivation and disability; educational skills and training; barriers to housing and services; crime; and living environment.27 The index covers an average population of 1500.27
For all 3 jurisdictions, we mapped the area-level deprivation indicator to the maternal postal code or postcode at delivery. To ensure relative comparability, we coded each indicator into jurisdiction-specific quintiles based on the distribution across the whole population.
Statistical analysis
We calculated all-cause mortality rates and rates of RTI-related mortality per 100 000 child-years overall and according to each risk factor. We constructed Kaplan–Meier plots to visualize unadjusted survival curves for RTI-related mortality according to area-level deprivation. We used Cox proportional hazards regression models to estimate hazard ratios (HRs) and 95% confidence intervals (CIs). We included gestational age, sex and the presence of high-risk chronic conditions (recorded before 1 or 5 yr of age), as well as the deprivation indicators (area-level deprivation and maternal age). We carried out Cox proportional hazards modelling separately for children aged 28–364 days and 1–4 years. We assessed the proportionality of hazards between deprivation groups by testing and inspecting Schoenfeld residuals after Cox model fitting. Statistical analyses were carried out with Stata version 15 (StataCorp) for England and Scotland, and SAS version 9.4 (SAS Institute) for Ontario.
Ethics approval
All analyses were run in parallel for each jurisdiction, as privacy legislation did not allow for the sharing or combining of individual-level patient data. Ethics approval was obtained through the Research Ethics Board at The Hospital for Sick Children, Toronto, and the Public Benefit and Privacy Panel for Scotland. The English data were exempt from English research ethics approval because the study involved analysis of an existing pseudonymized data set.28
Results
A total of 1 299 240 (Ontario), 547 556 (Scotland) and 3 910 401 (England) children were included in the study. The distribution of sex, gestational age and maternal age were broadly similar across jurisdictions (Table 1). Ontario had a higher proportion of early-term births (26.1% v. 16.2% in Scotland and 18.5% in England), and Scotland had a higher proportion of teenage mothers (4.1% v. 3.6% in England and 2.1% in Ontario). England had the highest proportion of children recorded as having high-risk chronic conditions, and Ontario had the lowest.
Selected risk factors for death related to respiratory tract infection among children born in Ontario, Scotland and England, Dec. 4, 2002, to Dec. 4, 2012
There were 2065 all-cause deaths among children less than 5 years of age in Ontario, 1091 in Scotland and 7195 in England. In all, 309 children in Ontario, 196 children in Scotland, and 1647 children in England died from an RTI, accounting for 15.0%, 18.0% and 22.9%, respectively, of all-cause deaths in children in this age group. Over the 10-year study period, both overall mortality rates and rates of RTI-related mortality were lowest in Ontario (Appendix 1, Supplemental Tables S4 and S5). In all 3 jurisdictions, more than 60% of all-cause deaths and 80% of RTI-related deaths among children aged 28 days to less than 5 years occurred in those with at least 1 high-risk chronic condition.
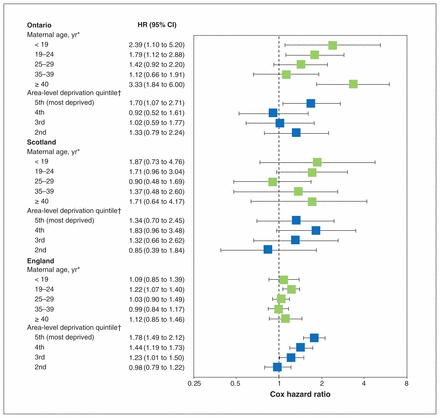
The adjusted Cox HRs for RTI-related death among children aged 28–364 days are presented in Figure 1 and Appendix 1, Supplemental Table S6. Independent of birth characteristics and deprivation quintile, children born to teenaged girls were at increased risk for RTI-related death in Ontario but not in Scotland or England. Independent of birth characteristics and maternal age, children born in the most deprived areas had higher rates of RTI-related death than those in the least deprived areas in all jurisdictions; however, this difference was not statistically significant for Scotland.
Adjusted Cox proportional hazard ratios (HRs) for mortality related to respiratory tract infection (RTI) among children aged 28–364 days, by jurisdiction. Adjusted for sex, gestational age, presence of high-risk chronic conditions, maternal age and deprivation quintile. Based on 170 RTI-related deaths in Ontario, 107 in Scotland and 961 in England. Error bars represent 95% confidence intervals (CIs). *Reference: 30–34 years. †Reference: 1st quintile (least deprived).
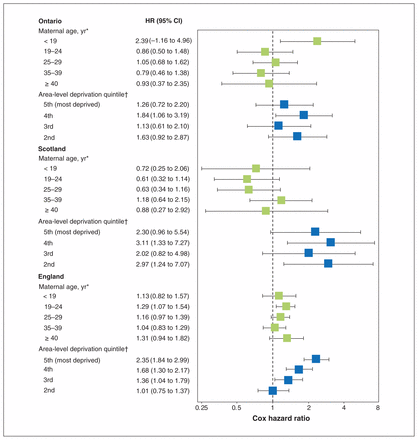
The adjusted HRs for RTI-related mortality for children aged 1–4 years are shown in Figure 2 and Appendix 1, Supplemental Table S7. Again, children born to teenaged girls were at increased risk for RTI-related death in Ontario but not in England or Scotland. In contrast, children born in more deprived areas were at increased risk compared to those born in the least deprived areas in England and Scotland but not in Ontario.
Adjusted Cox proportional hazard ratios (HRs) for mortality related to respiratory tract infection (RTI) among children aged 1–4 years, by jurisdiction. Adjusted for sex, gestational age, presence of high-risk chronic conditions, maternal age and deprivation quintile. Based on 139 RTI-related deaths in Ontario, 89 in Scotland and 686 in England. Error bars represent 95% confidence intervals (CIs). *Reference: 30–34 years. †Reference: 1st quintile (least deprived).
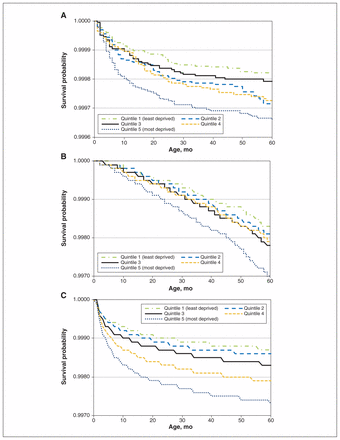
Across all jurisdictions, children born in more deprived areas experienced increasingly higher rates of RTI-related mortality (Figure 3). There was evidence that the effect of deprivation stratified in the 2 age groups (28–364 d and 1–4 yr) was not proportional in Scotland and Ontario but appeared proportional in England.
Kaplan–Meier survival curves for mortality related to respiratory tract infection (RTI), by area-level deprivation quintile for Ontario (A) (n = 1 299 240; 2065 all-cause deaths; 309 RTI-related deaths), Scotland (B) (n = 547 556; 1091 all-cause deaths; 196 RTI-related deaths) and England (C) (n = 3 910 401; 7195 all-cause deaths; 1647 RTI-related deaths). y axes are shown on different scales.
Interpretation
We identified area-level socioeconomic disparities in the risk of RTI-related death among children in England and Ontario, 2 high-income jurisdictions with universal health care programs, even after adjustment for risk factors such as prematurity and high-risk chronic conditions.
Teenage motherhood was a significant risk factor among children born in Ontario but not in England or Scotland. We hypothesize that this may be due to differences in support systems for young mothers24,29,30 and may also reflect higher rates of teenage pregnancy, along with increased all-cause infant mortality rates, in Indigenous populations.31 In a study using data from 2002–2006, the rate of teenage motherhood was more than 5 times greater among Indigenous women than non-Indigenous women in Canada, and the rate of all-cause infant mortality was more than 2 times greater;31 however, to our knowledge, the combination of these 2 factors has not been explored. Indigenous families in Canada are also more likely to have other risk factors for childhood death related to RTI, such as poor-quality or crowded housing.31,32 These results highlight the importance of considering context-specific differences in potential mechanisms underlying socioeconomic inequities in pediatric health outcomes, particularly health-care–amenable conditions, such as RTI-related death.
This study adds to a rich body of literature showing that socioeconomic disparities in amenable pediatric mortality continue to exist.18,30,33,34 It also adds to a smaller body of literature providing standardized, cross-jurisdictional comparisons of disparities in childhood mortality. Notably, Zylbersztejn and colleagues18 used a similar standardized approach to compare childhood mortality in England and Sweden and found stark differences in mortality patterns between the 2 countries; these differences were largely associated with variations in socioeconomic factors, particularly those that increase the risk of adverse birth outcomes, such as low birth weight. Like us, they observed that the relative importance of various socioeconomic factors, including maternal age and socioeconomic status quintile, varied in each of these countries.
Further work is needed to determine how socioeconomic status may affect access to preventive interventions, such as vaccination, and timely medical care. Our findings also highlight the necessity of understanding context-specific drivers of socioeconomic disparities in health outcomes.35 For example, we identified young maternal age as being particularly associated with increased risk for childhood mortality in Ontario. A more detailed analysis of the factors underlying this association in Ontario would be needed to better understand this association, including any potential modification by other factors, such as being an Indigenous person.
Limitations
Since death from RTI is rare, a population-based analysis including large numbers of children was required. Although this method allowed for the identification of RTI-related deaths, it was not possible to determine whether a child died directly as a result of an RTI.10,36 Similarly, owing to variability in and undercoding of specific causes of RTI-related death, we were unable to accurately investigate deaths due to specific pathogens.
We used different area-level measures of socioeconomic deprivation in each jurisdiction, since individual-level data on maternal education or family income were not available across the 3 jurisdictions. Although each measure conceptualizes socioeconomic deprivation, they may measure slightly different constructs and provide different levels of precision, which likely blunted associations. This may explain why, in contrast to Ontario and England, we observed no statistically significant association between area-level deprivation and RTI-related mortality in Scotland. Nonetheless, given that quintile measures are compared, each allows for the relative comparison of social groups within a jurisdiction.
Furthermore, we could not examine which aspects of deprivation, such as maternal education (associated with better access to childhood vaccination services, for example37,38), overcrowded or damp housing,39 and exposure to air pollution,40 were the most important in explaining within-jurisdiction socioeconomic status gradients in RTI-related mortality. In particular, exposure to tobacco smoke during pregnancy has been reported to differ among the 3 jurisdictions: 9.2% of pregnant women in Ontario smoked tobacco cigarettes in 2010,41 compared to recent estimates of 18.8% in Scotland42 and 13.6% in England.43 Similarly, there were several covariates that could not be included, such as parental health status, propensity to seek health care and Indigenous status (in Canada). Thus, there is a risk of residual confounding. This likely would result in underestimation of the effect of socioeconomic disparities on child health, as many confounders would be distally associated with lower socioeconomic status.
We found evidence to suggest that the effects of deprivation were not proportional within the 2 age groups. We decided against including time interactions to account for nonproportional hazards, as these would differ by jurisdiction and hinder comparability of the results. Qualitatively, evidence of nonproportional hazards further emphasizes that the effect of social inequities plays out differently in each jurisdiction.
Critically, individual-level vaccination status was not available in all the jurisdictions included in this study. According to ecologic estimates (Appendix 1, Supplemental Table S1), vaccination coverage for common RTIs varied notably across jurisdictions, despite being publicly funded.44,45 This may explain some of the variability between jurisdictions in rates and trends of death from RTI; however, although Ontario generally had the lowest vaccination coverage rates, rates of RTI-related death were lowest in that jurisdiction.
Although hospital diagnosis codes generally have high sensitivity and specificity,46 the specific codes used to identify high-risk chronic conditions, based on an existing categorization of pediatric chronic conditions,23 have not been validated. Thus, there is potential for misclassification, the magnitude of which may vary across jurisdictions given differences in coding practices, diagnostic guidelines, and funding incentives based on comorbidity or diagnostic coding accuracy. We also assumed that children were biologically at risk during the whole study period, and, thus, we did not consider the time-dependent nature of some deaths from these conditions in our analysis. Although this is a limitation of our analysis for any condition that is time-dependent (e.g., immunosuppressed states that would only follow cancer treatment), most (80%) of the high-risk chronic conditions that occurred in the children included in this study were diagnosed by the time of their first birthday. Thus, the analysis of children aged 1–4 years is approximately correct.
Finally, censorship due to outmigration varied across jurisdictions. As data updates on outmigrated children require interaction with the health and social care systems, outmigration was likely underestimated in our study. Depending on the health status of outmigrating children, this may have biased the association between socioeconomic status and RTI-related mortality in either direction.
Conclusion
In children who survived to age 28 days, socioeconomic disparity predicted the likelihood of RTI-related death. This relation was particularly apparent in Ontario and England, where area-level deprivation remained associated with RTI-related mortality even after birth characteristics such as presence of high-risk chronic conditions and prematurity were accounted for. Teenage motherhood conferred a higher risk of crude mortality across all 3 jurisdictions, and, in Ontario, this also persisted as an independent risk factor for RTI-related death. These results highlight the need for further, context-specific investigation around the mechanisms of this increased risk and the development of programs to address these disparities, including an increased focus on ensuring that available programs reach children at higher risk.
Acknowledgements
The authors thank members of the Children and Families Policy Research Unit, particularly Profs. Catherine Law and Terence Stephenson, for their support. They also acknowledge support from the National Institute for Health Research Biomedical Research Centre at Great Ormond Street Hospital for Children NHS Foundation Trust and University College London. The authors thank Katie Harron and Linda Wijlaars for their help with mother–baby linkage in the England cohort, and Carole Morris for her help with the Scottish birth cohort.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Laura Holder created the birth cohort for Ontario, and Ania Zylbersztejn created the birth cohort for England. Maximiliane Verfürden and Pia Hardelid analyzed the data. Tiffany Fitzpatrick coordinated the Ontario analysis under the supervision of Astrid Guttmann. Maximiliane Verfürden, Pia Hardelid and Tiffany Fitzpatrick drafted the manuscript. Astrid Guttmann, Laura Rosella and Ruth Gilbert revised the manuscript critically for important intellectual content. All of the authors conceptualized and designed the study, approved the final version to be published and agreed to be accountable for all aspects of the work. Astrid Guttmann and Pia Hardelid are joint senior authors. Maximiliane Verfürden and Tiffany Fitzpatrick are joint first authors.
Funding: The Ontario analysis was supported by competitive operating grant FRN-142498 to Laura Rosella from the Canadian Institutes of Health Research (CIHR). This study was also supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care. Parts of this material are based on Ontario Registrar General information on deaths, the original source of which is ServiceOntario, as well as on data and information compiled and provided by the Canadian Institute for Health Information. Astrid Guttmann was supported by a CIHR Applied Chair in Reproductive and Child Health Services and Policy Research (APR 126 377), which also provided some of the operating funding for this study. Pia Hardelid was funded by postdoctoral fellowship PDF-2013-06-004 from the National Institute for Health Research (NIHR). This article represents independent research funded by the NIHR. This study contributed to responsive research on mortality amenable to health care for the Children and Families Policy Research Unit (reference 109/00017), which is funded by the Department of Health Policy Research Programme at University College London. This is an independent report commissioned and funded by the UK Department of Health. The study benefited from infrastructure provided by the NIHR Great Ormond Street Hospital Biomedical Research Centre. Ruth Gilbert receives funding from Health Data Research UK.
Data sharing: These data were reused with the permission of The Health and Social Care Information Centre (now NHS Digital) (NIC-393510-DH1D). All rights reserved. Copyright © 2017. The data set from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the dataset publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS. The full data set creation plan and underlying analytic code are available from the authors on request, with the understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore inaccessible or may require modification.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources; no endorsement by ICES, the Ontario MOHLTC, the Canadian Institute for Health Information, ServiceOntario or the Canadian Institutes of Health Research is intended or should be inferred. The views expressed are those of the authors and not those of the National Health Service, the National Institute for Health Research or the Department of Health.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/8/2/E273/suppl/DC1.
References
- Copyright 2020, Joule Inc. or its licensors