Abstract
Background: The importance of sex and gender in the diagnosis and management of health conditions is well established, but the extent to which this evidence is integrated into clinical practice guidelines remains unknown. We aimed to determine the proportion of Canadian clinical practice guidelines that integrate evidence on sex and gender considerations.
Methods: We searched the Canadian Medical Association's CPG Infobase, PubMed, all provincial/territorial websites and websites of professional organizations for English- and French-language Canadian clinical practice guidelines published between January 2013 and June 2015 on selected conditions identified as priorities by policy-makers and practitioners. Citations and text were searched electronically using keyword terms related to sex and gender. Three investigators independently analyzed and categorized the content of text-positive clinical practice guidelines based on clinical relevance for practitioners.
Results: Of the 118 clinical practice guidelines that met the inclusion criteria, 79 (66.9%) were text-positive for sex and/or gender keywords; 8 (10%) of the 79 used the keywords only in relation to pregnancy. Of the remaining 71 guidelines, 25 (35%) contained sex-related diagnostic or management recommendations. An additional 5 (7%) contained recommendations for sex-specific laboratory reference values, 29 (41%) referred to differences in epidemiologic features or risk factors only, and 12 (17%) contained nonrelevant mentions of search keywords. Twenty-five (35%) of the text-positive guidelines used the terms "sex" and/or "gender" correctly.
Interpretation: Recommendations related to sex and gender are inconsistently reported in Canadian clinical practice guidelines. Guidelines such as the Sex and Gender Equity in Research guidelines may help inform the meaningful inclusion of sex and gender evidence in the development of clinical practice guidelines.
Manifestations and outcomes of disease, including cardiovascular conditions,1-4 immune disease,5,6 respiratory illness6-8 and mental health conditions,9,10 have long been known to differ between men and women. These differences relate to sex (molecular, cellular and epigenetic mechanisms of male and female physiology) and gender (adopted or imposed social norms, behaviours, identities and expectations).11,12 Better understanding of these mechanisms has led to a near-linear rise in sex- and gender-specific research publications since the 1990s in all medical disciplines,13 although terminology can be confused.11
Despite new evidence on differences between males and females, the uptake of sex and gender influences into clinical practice guidelines and clinical practice has been slow.14 The development of clinical practice guidelines is a critical first step for translating research findings into clinical practice to narrow the "know-do" gap15,16 and improve patient care. The consequences of not including evidence about females and males separately in clinical practice guidelines can range from missed opportunities to prevent type 2 diabetes in fathers of children whose mothers had gestational diabetes17 to inappropriate prescription of certain cardiovascular drugs to women based on altered risk-benefit profiles.18 For example, in 2014, Health Canada issued a warning to cut the recommended dosage of zolpidem, a common sleeping pill, for women by half because morning blood levels of the drug are higher in women than in men, with increased risk of driving impairment.19 Rochon and colleagues20 discovered that, in Ontario, men with dementia who were prescribed antipsychotic drugs had significantly higher risks for hospital admission and death than did women with dementia. Similarly, social risk factors are associated with higher rates of suicide in older men, which suggests that more aggressive screening and treatment may be required.21-23
The extent to which evidence about sex and gender is integrated into clinical practice guidelines for diagnosis and management of diseases remains unknown. We conducted a systematic review to investigate the integration of sex and gender evidence into Canadian clinical practice guidelines published between 2013 and 2015 for noncommunicable health conditions.
Methods
Data sources and inclusion criteria
Protocols for this review are posted on Open Science Framework and follow PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) reporting guidelines (www.prisma-statement.org/). The review search focused on selected health conditions identified as priorities by policy-makers and practitioners24-30 (Box 1). We considered only 2 cancers, lung and colorectal, because they are among the leading causes of death from cancer for both females and males,31 and important sex and/or gender differences in prevalence, risk and screening, pathophysiologic characteristics, diagnosis, treatment and outcomes for these conditions have been found.32-36
Health conditions identified as priorities by policy-makers and practitioners24-30 and included in the search for clinical practical guidelines
Acute coronary syndrome
Addictions
Alcohol drinking
Angina pectoris
Anxiety disorders
Asthma
Atrial fibrillation
Back pain
Cancer, colorectal
Cancer, general
Cancer, lung
Cardiomyopathies
Cardiovascular diseases
Chronic obstructive pulmonary disease
Coronary artery disease
Dementia
Depression
Diabetes
Diabetic nephropathies
Dyslipidemias
Emphysema
Fibromyalgia
Generalized anxiety disorder
Heart failure
Hypertension
Insomnia
Mental health conditions
Migraine
Myocardial ischemia
Neuralgia
Obesity
Osteoarthritis
Pain
Peripheral vascular diseases
Polyps
Pulmonary embolism
Pulmonary hypertension
Stroke
Substance dependence
Thrombosis
Transient ischemic attacks
Venous thromboembolism
We searched the Canadian Medical Association's database (CPG Infobase) of about 1200 clinical practice guidelines developed or endorsed by authoritative medical or health organizations in Canada37 for guidelines related to the identified health conditions. We also searched PubMed, all Canadian provincial/territorial websites and professional association websites for English- and French-language clinical practice guidelines using the search term "clinical practice guideline." Only documents published in Canada between January 2013 and June 15, 2015 were included. We chose these dates because Health Canada's Health Portfolio Sex and Gender-Based Analysis Policy38 was issued in 2009; new research under the policy would be published 1 to 2 years later, and at least 2 more years are required to review evidence and develop a clinical practice guideline.16 We excluded original research, reviews, opinion pieces and editorials as well as clinical practice guidelines for single-sex health conditions such as menopause and prostate or gynecologic cancers. Duplicates were removed.
Data extraction and quality assessment
We searched the text and citations of clinical practice guidelines electronically for the keywords "sex," "gender," "male," "female," "men," "women," "man," "woman," "boy," "girl" and "pregnan*." We searched for clinical practice guidelines in French using the keywords "sexe," "genre," "sexo-specifique," "homme," "hommes," "femme," "femmes," "fille," "filles," "garçon," "garçons" and "grossesse." We did not perform risk-of-bias assessments, as there is no validated strategy for sex and gender integration in clinical practice guidelines. Guidelines were considered text-positive if they had any keywords in the main text and text-negative if they had no keywords in the main text.
Data synthesis and analysis
We assigned guidelines that covered more than 1 health condition to the condition most emphasized in the text. Text-positive and text-negative guidelines were treated separately. Three investigators (C.T., B.C. and M.H.-B.) independently analyzed the content of every text-positive guideline for the type, amount and applicability of evidence presented on epidemiologic features, risk and screening, pathophysiologic characteristics, symptoms and diagnosis, and treatment/interventions.14
We assigned text-positive guidelines to 1 of 5 categories according to the relevance of the information to clinical practice. Guidelines were assigned to category 1 if evidence-informed recommendations were made that supported different approaches for men and women, and to category 2 if the evidence presented supported the use of a singular approach for women and men. Category 3 included guidelines with sex-specific reference values for laboratory data. Guidelines that reported sex or gender differences in epidemiologic features or risk factors but did not make suggestions for clinical management were assigned to category 4. Category 5 was reserved for guidelines that made only superficial mention of 1 or more of the search terms. The use of "sex" and "gender" specifically was assessed for adherence to current definitions.11,38 Differences in categorization were resolved by consensus.
Outcomes
The primary outcome of the review was the proportion of clinical practice guidelines that mentioned sex or gender or both, defined as the number of guidelines that were text-positive for any keyword over the total number of guidelines included in the review. Secondary outcomes were a qualitative analysis39 of whether the inclusion of evidence in the text was clinically relevant, and the appropriate use of the terms "sex" and "gender" according to the Sex and Gender Equity in Research guidelines.11
Statistical analysis
We did not conduct statistical analyses of guideline data as the intent of this review was strictly to identify and categorize any mentions of sex- or gender-related words in the clinical practice guideline texts.
Results
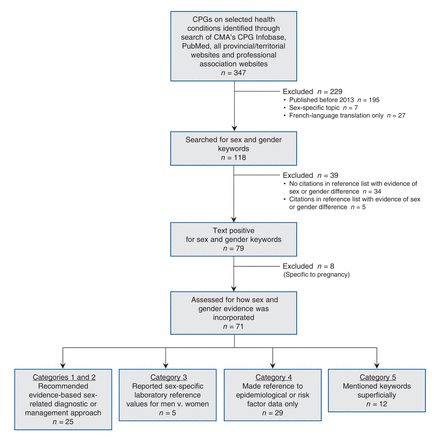
The search identified 347 Canadian clinical practice guidelines that addressed 1 or more of the health conditions under consideration. We excluded 229 guidelines because of publication before 2013, a sex-specific topic or duplication, yielding 118 guidelines for final inclusion in the analysis (Figure 1), 3 of which were in French only.
Flowchart showing the inclusion and exclusion criteria that were applied to the initial sample of 347 Canadian clinical practice guidelines on selected health conditions. A total of 71 text-positive guidelines were categorized as a function of the clinical relevance of the information provided. CMA = Canadian Medical Association, CPG = clinical practice guideline.
Inclusion of sex and/or gender information
Of the 118 clinical practice guidelines, 79 (66.9%) were text-positive for sex and gender keywords. Eight of the 79 (10%) used keywords only in relation to pregnancy. Of the 71 remaining guidelines, 25 (35%) contained sex-related diagnostic or management recommendations; 21 (30%) recommended different approaches for men and women, and 4 (6%) recommended similar approaches based on evidence (categories 1 and 2, respectively). Five guidelines (7%) included recommendations for sex-specific laboratory reference values (category 3). Twenty-nine guidelines (41%) referred to differences in epidemiologic features or risk factors only (category 4), and 12 (17%) contained mentions not relevant to clinical practice (category 5).
Of the 39 text-negative guidelines, 5 (13%) contained citations in the reference lists alluding to the presence of sex differences in the condition of interest. For example, a guideline on the pharmacological management of chronic neuropathic pain contained no keywords in the body of the text but cited 2 articles on the effects of sustained-action opioids on women and men.40
Type of sex and gender evidence cited in text-positive clinical practice guidelines
The nature of sex-specific information reported in text-positive clinical practice guidelines varied (Table 1). For example, 1 guideline stated that "colorectal cancer is the third most common cancer … in both sexes,"68 whereas a few provided sex-disaggregated risk and mortality data, such as "The probability of developing colorectal cancer increases with age and varies with sex … i.e., 1/32 males and 1/36 females will die of invasive colorectal cancer."69,70
Other guidelines mentioned sex as a risk factor but only in absolute terms, such as noting that female sex is a risk factor for depression in patients with diabetes71 or that female adolescents with type 1 diabetes are at risk for eating disorders.72 Sometimes the comparative risk of different outcomes in men and women was mentioned in broad terms, such as following stroke, when women are at greater risk for depression whereas men are at greater risk for vascular dementia.73 In rare instances, sex differences in risk were explicit: "Estimates of risk of ischemic stroke in people with diabetes range from a 2- to 3-fold increase in men and a 2- to 5-fold increase in women."74 In only 2 cases were gender differences noted, as in the case of perceptions and interpretations of pain.67
Some guidelines provided evidence that can inform clinical practice. For example, guidelines on alcohol consumption recommended a lower diagnostic screening threshold for risky drinking behaviour for women than for men,53,54,75 although only 1 guideline explained biological differences in alcohol absorption rates by sex.53 Another guideline suggested that women respond more favourably than men to outpatient care.75 Two guidelines alluded to hormonal causal pathways for aggravated hypertension in women, highlighting modifiable treatment strategies in the form of discontinuation of oral contraceptive and exogenous sex hormone therapies.45,76 A recommendation to avoid antidepressants with strong cytochrome P450 2D6 inhibition (e.g., paroxetine, fluoxetine) in premenopausal survivors of breast cancer requiring tamoxifen therapy can be applied in practice.77 As well, a guideline on heart failure in children and youth warned that treatment with spironolactone can lead to irreversible gynecomastia in males, affecting long-term psychological outcomes.78
Conversely, some guidelines did not address the clinical implications of sex and gender differences. A guideline noting differences in response to medication between male and female children and adolescents with type 2 diabetes did not recommend distinct treatment choices.72 Similarly, guidelines that described a higher rate of adverse effects of statin use in women than in men did not offer assistance for selecting alternative therapies to lower lipids.59,79,80
Uses of "sex"and "gender" evidence in clinical practice guidelines
Text-positive clinical practice guidelines also varied in the correct use of the terms "sex" and "gender." Of the 79 guidelines, 37 (47%) did not use either term explicitly, 14 (18%) used one or the other, but not according to the accepted definitions, and 25 (32%) used at least 1 term appropriately. One guideline (1%) used "sex" in the context of sexual activity only.
Interpretation
We found inconsistencies in the extent and degree to which evidence on sex and gender is incorporated into Canadian clinical practice guidelines. Only 35% of guidelines reported screening, diagnosis or management considerations specific to sex or gender, and only 25% used the terms "sex" and "gender" correctly. Given early recognition of sex and gender differences in mental health conditions and cardiovascular disease, we expected clinical practice guidelines in these fields to reliably highlight information about females and males separately.13 Recent attention to sex differences in cancer,81-83 lung disease6-8 and diabetes84-86 predicted meaningful uptake in these areas as well. Although some clinical practice guidelines targeting these conditions incorporated differentiated data in a way that guides practice, many keyword mentions were superficial and of limited value.
These findings are similar to those reported from the Netherlands, where a preliminary study showed that sex-related factors or effects were mentioned in only 20% of clinical practice guideline recommendations for osteoporosis and were completely absent in clinical practice guidelines on depression.87
Several reasons may account for the variable inclusion of disaggregated evidence in clinical practice guidelines. A simple explanation is the lack of research on sex and gender differences in medicine. With most biomedical experiments conducted exclusively on male animals and substantial underrepresentation of women in clinical trials, it is possible that many sex and gender differences have yet to be discovered.88,89 Although more publications that consider females and males separately in medicine are emerging, less than 25% address differences in management decisions for patients based on sex or gender.13
Another explanation for the suboptimal integration of sex and gender considerations into clinical practice guidelines relates to the lag time between research discovery and practice transformation.90,91 A recent study showed that microglial cells play a role in mediating pain pathways in male mice, whereas T cells are responsible for this same function in female mice.92 It is not known when and how this information will translate into different analgesic drug targets for men and women. Pharmacoepidemiological findings and postmarketing drug safety warnings on the differential risks of adverse drug reactions in women and men do not seem to substantially change entrenched prescribing patterns.93 Future clinical practice guidelines on insomnia should incorporate Health Canada's 2014 recommendation to cut the dosage of sleeping pills in half for women.19
Finally, failure to integrate such considerations into clinical practice guidelines likely results from lack of awareness as well as lack of guidance on how to do so.87,94,95 Current guideline development instruments do not provide instruction for synthesizing sex and gender evidence.96 The AGREE (Appraisal of Guidelines, Research and Evaluation) II instrument requires systematic reviewers to specify populations of interest, but does not require identification of evidence particular to males or females or any note of sex or gender differences.97 The same is true for GRADE (Grading of Recommendations Assessment, Development and Evaluation) guidelines.98-100 Unless a clinical practice guideline working group specifically asks a question about evidence-based sex and gender differences, it is unlikely that the correct search terms will be used to query the literature.
Several instruments exist to guide the inclusion of sex and gender. The Health Equity Group at Cochrane Canada encourages systematic reviewers to consider sex, gender and other equity dimensions, providing user templates.101 The Sex and Gender Equity in Research guidelines provide rationales and guidance for clinical practice guideline reviewers to look for and incorporate evidence on sex differences and gender and to use the terms appropriately.11,102 Song and colleagues103 published a validated search strategy for uncovering publications that report sex-specific diagnostic and treatment evidence. Standardized application of these strategies could enhance the uptake of evidence that is sex and gender specific in future clinical practice guidelines.
Limitations
A strength of our systematic review is that we used Canadian, provincial and academic Web sources to search for clinical practice guidelines, and the search terms were broad and inclusive. One limitation is that our findings risk overestimating perceptions that sex and gender evidence is meaningfully integrated into Canadian clinical practice guidelines. Ultimately, only 25 (21.2%) of the 118 clinical practice guidelines included in our review provided relevant sex-related diagnostic or management recommendations. We did not perform risk-of-bias assessments of the quality of the guidelines. A systematic search for sex- and gender-specific evidence that was omitted from clinical practice guidelines would permit an assessment of any missed opportunities to appropriately include information about male and female patients in clinical practice guidelines.
Conclusion
Gender-responsive health care takes into account all sex- and gender-related factors for women and men. Practitioners are increasingly requiring male- and female-specific information. For example, specialists in heart failure have explicitly called for sex-specific diagnostic criteria and treatments. The implications of this systematic review on future clinical practice guideline development are twofold. First, we suggest that guidelines for developing clinical practice guidelines be revised to emphasize the importance of including subquestions and search strategies that reveal evidence on sex and gender differences in medicine. Second, we recommend that a focus on sex- and gender-responsive interventions be prioritized not only in clinical practice guidelines but also in medical school curricula and continuing education to consolidate Canadian innovation in personalized care.
Acknowledgements
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/5/1/E66/suppl/DC1
Footnotes
Competing interests: Barbara Clow and Margaret Haworth-Brockman received consulting fees from the Institute of Gender and Health, Canadian Institutes of Health Research during the conduct of the study.
Contributors: Barbara Clow and Margaret Haworth-Brockman performed the search for clinical practice guidelines and searched the guidelines for sex and gender keywords. Cara Tannenbaum, Barbara Clow and Margaret Haworth-Brockman analyzed and categorized the guidelines. All of the authors drafted the manuscript, revised it critically, approved the final version to be published and agreed to act as guarantors of the work.
References
- Copyright 2017, Joule Inc. or its licensors