Abstract
Background: Evidence-based Practice for Improving Quality (EPIQ) is a collaborative quality improvement method adopted by the Canadian Neonatal Network that led to decreased mortality and morbidity in very preterm neonates. The Alberta Collaborative Quality Improvement Strategies to Improve Outcomes of Moderate and Late Preterm Infants (ABC-QI) Trial aims to evaluate the impact of EPIQ collaborative quality improvement strategies in moderate and late preterm neonates in Alberta, Canada.
Methods: In a 4-year, multicentre, stepped-wedge cluster randomized trial involving 12 neonatal intensive care units (NICUs), we will collect baseline data with the current practices in the first year (all NICUs in the control arm). Four NICUs will transition to the intervention arm at the end of each year, with 1 year of follow-up after the last group transitions to the intervention arm. Neonates born at 32 + 0 to 36 + 6 weeks’ gestation with primary admission to NICUs or postpartum units will be included. The intervention includes implementation of respiratory and nutritional care bundles using EPIQ strategies, including quality improvement team building, quality improvement education, bundle implementation, quality improvement mentoring and collaborative networking. The primary outcome is length of hospital stay; secondary outcomes include health care costs and short-term clinical outcomes. Neonatal intensive care unit staff will complete a survey in the first year to assess quality improvement culture in each unit, and a sample will be interviewed 1 year after implementation in each unit to evaluate the implementation process.
Interpretation: The ABC-QI Trial will assess whether collaborative quality improvement strategies affect length of stay in moderate and late preterm neonates. It will provide detailed population-based data to support future research, benchmarking and quality improvement.
Trial registration: ClinicalTrials.gov, no. NCT05231200
Preterm birth represents 8% of live births in Canada. Of these, 85% are moderate and late preterm neonates born at 32–36 weeks’ gestation.1 These neonates are at an increased risk of serious morbidities and prolonged hospitalizations,2,3 and the families of these neonates often face considerable financial and psychosocial burdens.4–7 Practice variation in care of moderate and late preterm neonates is associated with inconsistent outcomes and variable length of stay between neonatal intensive care units (NICUs).8–10
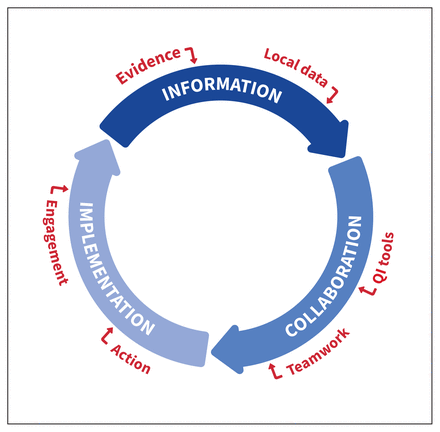
Collaborative quality improvement is an organized, multifaceted approach that includes teams from multiple health care sites coming together to learn, apply and share improvement methods, ideas and data on service performance for a given health care topic. In a systematic review of 64 studies, applying collaborative quality improvement practices led to significant improvements in clinical processes and patient outcomes.11 Studies in preterm neonates born at less than 32 weeks using Evidence-based Practice for Improving Quality (EPIQ),12–14 a collaborative quality improvement methodology adopted by the Canadian Neonatal Network,15 decreased mortality and morbidity in neonates born at less than 29 weeks’ gestation.16 The EPIQ quality improvement cycle is driven by information, collaboration and implementation (Figure 1).17 This is based on the Promoting Action on Research Implementation in Health Services framework for conceptualizing successful implementation of evidence-based practices.18 Although collaborative quality improvement interventions can be associated with upfront costs, including infrastructure and personnel, a systematic review showed the potential of collaborative quality improvement for cost saving for both chronic and acute conditions. 19 In high-risk neonates, collaborative quality improvement was associated with cost savings that offset the institutional expenditure for its activities.20
The EPIQ cycle is based on principles of information, collaboration and implementation. Used with permission from the Canadian Neonatal Foundation. Note: EPIQ = Evidence-based Practice for Improving Quality, QI = quality improvement.
Despite the high proportion of moderate and late preterm neonates among NICU admissions, they are underrepresented in research and quality improvement activities, resulting in a lack of detailed population-based data for research, benchmarking and quality improvement. The ABC-QI Trial aims to evaluate the impact of collaborative quality improvement interventions in moderate and late preterm neonates on length of stay, clinical outcomes and cost to the health care system in 12 NICUs across Alberta, Canada.
Methods
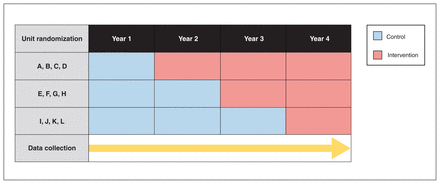
This study is a multicentre, stepped-wedge cluster randomized trial that involves 12 NICUs across the province of Alberta. There are 14 NICUs in Alberta: 10 level II units, where generally admission criteria include gestational age 32 weeks or greater; 2 perinatal level III units (Foothills Medical Centre in Calgary and Royal Alexandra Hospital in Edmonton); 1 level III surgical NICU at Alberta Children’s Hospital; and 1 level IV NICU at Stollery Children’s Hospital. The latter 2 NICUs will not participate in the study as most patients admitted require surgical interventions. See Appendix 1, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1, for details of Alberta NICUs. Each of the 12 participating NICUs is considered a cluster and will be randomized individually to transition from the control arm (current practice) to the intervention arm (collaborative quality improvement strategies) at 3 time points while stratifying by location, urban versus regional, and level of care (Figure 2). Randomization will be conducted 9 months after initiation of data collection.
ABC-QI trial stepped-wedge cluster randomization design and timeline. In the first year, all 12 participating neonatal intensive care units (NICUs) will be in the control arm. Each year, 4 NICUs will be randomized to transition to the intervention arm. At year 4, all participating NICUs will be in the intervention arm. Data will be collected from neonates (length of stay and clinical outcomes), for costs to health care system per patient, and from staff (perceptions of collaborative quality improvement implementation). Note: ABC-QI = Alberta Collaborative Quality Improvement Strategies to Improve Outcomes of Moderate and Late Preterm Infants.
The advantages of stepped-wedge cluster randomized trial design compared with parallel cluster randomized trial design include the following: ability to conduct randomized evaluation of system interventions that are frequently rolled out sequentially without robust evaluation;21 facilitating cluster recruitment as the participating units were hesitant to enroll in a parallel cluster randomized trial; the logistic feasibility of implementing a complex collaborative quality improvement intervention sequentially rather than en bloc;22 achieving higher power compared with parallel cluster randomized trial with the similar number of clusters;21 and aligning with the 6 dimensions of Alberta Quality Matrix for Health (acceptability, accessibility, appropriateness, effectiveness, efficiency and safety).23 There are risks associated with the stepped-wedge cluster randomized trial design, including implementing an intervention, with unproven effectiveness, in all clusters. 24 However, stepped-wedge cluster randomized trial design is used in implementation studies of proven interventions, 25 such as the previously validated EPIQ collaborative quality improvement methodology.
Study population
Using a waiver of consent, all neonates born at 32 + 0 to 36 + 6 weeks’ gestation and admitted to participating NICUs or postpartum units between Sept. 1, 2022, and Aug. 31, 2026, will be included. Exclusion criteria include neonates with major congenital anomalies or chromosomal abnormalities (Appendix 2, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1), primary admission to a surgical NICU, and neonates born in or transferred to a NICU outside of Alberta.
Staff participants
Management, nurses, physicians and other health care providers will be invited by email to participate in the Quality Improvement Implementation Survey version 2 (QIIS-II). Posters in staff areas and emails from managers will facilitate the recruitment of participants for the semistructured interviews.
Randomization
Although full stratification by site characteristics is not possible with only 12 clusters, there are 2 goals of the randomization: to avoid having 2 level III NICUs in the same step and to ensure that none of the steps has more than 2 regional NICUs. Using R software, we developed an algorithm to accomplish these criteria (Appendix 3, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1).
Interventions
The study intervention consists of implementation of 2 standardized practice care bundles. A care bundle is a small, simple set (3–5 elements) of evidence-based practices that, when performed collectively and reliably, result in improved patient outcomes. 26 The process of care bundle development and refinement is described in Appendix 4 (available at www.cmajopen.ca/content/11/3/E397/suppl/DC1). We selected respiratory and nutritional and feeding management, as those are major predictors of length of stay.27,28
Respiratory care bundle: stabilization and respiratory care in moderate and late preterm neonates. 1) Establishing effective ventilation in the delivery room, 2) prevention of hypothermia, 3) early diagnosis and management of respiratory distress with continuous positive airway pressure, 4) standardized approach for surfactant indications and administration and 5) standardized approach for early extubation.
Nutritional and feeding care bundle: nutritional support in moderate and late preterm neonates. 1) Early initiation of enteral or parenteral nutrition, 2) standardized tables for feeding initiation and progression, 3) optimizing breastfeeding and use of breastmilk and 4) standardized approach for the transition from enteral nutrition via tube feeds to oral feeds.
To facilitate this implementation, the study team will use 5 collaborative quality improvement strategies.
Quality improvement team building: Each NICU will create a multidisciplinary quality improvement team, including a parent advisor when feasible, to lead the quality improvement activities and education, and champion the culture and practice change.
Quality improvement education: quality improvement teams will receive standardized quality improvement education using the 6-hour EPIQ Workshop,29 which involves a hands-on approach to enable successful implementation of quality improvement projects.30 EPIQ workshops will be delivered by the study team for quality improvement teams in each unit within 3 months before transition to the intervention arm, either in person or virtually, based on the public health guidance at the time.
Implementation of care bundles: local quality improvement teams will use EPIQ methods through engagement and education of front-line staff to create change ideas that are translated into the unit culture using EPIQ 10 steps, including Plan-Study-Do-Act cycles (Appendix 5, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1). The aim is to implement the 2 care bundles within 12 months of transition to the intervention arm. The type and number of Plan-Study-Do-Act cycles may vary between the units based on their current practice, previous and concurrent quality improvement activities, and educational needs.
Mentoring: each NICU will have 1 or more mentors from the study team who are experienced in EPIQ methods to help local quality improvement teams engage front-line staff in quality improvement and navigate the unit-specific challenges during the intervention.
Collaborative networking: NICUs in the intervention will participate in quarterly virtual meetings to discuss progress and share their Plan-Study-Do-Act cycles and data to learn from each other. Neonatal intensive care units will have access to their data and receive quarterly reports using statistical process control charts outlining their performance compared with the other units and the overall average.
Control arm (current practices)
All participating NICUs will be in the control arm during the first year to collect baseline data of the current practices and between-unit variation, prepare repositories of existing guidelines and survey existing quality improvement activities. Neonatal intensive care units in the control arm can continue conducting quality improvement activities relevant to their practice, but without receiving the interventions outlined above until they transition to the intervention arm. These activities will be captured and accounted for in the analysis.
Outcomes
The primary outcome is length of hospital stay. Secondary outcomes include cost to the health care system; and short-term clinical outcomes, including respiratory and nutritional care, weight, postdischarge information, mortality and transfers from level II to level III NICUs (Appendix 6, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1).
Data sources
The study database is built using REDCap (Research Electronic Data Capture) tools hosted at the University of Alberta.31,32 See Appendix 7 (available at www.cmajopen.ca/content/11/3/E397/suppl/DC1) for postdischarge data sources. The Canadian Institute for Health Information (CIHI) Patient Cost Estimator33 will be used to calculate daily cost. Data will be abstracted from paper and electronic patient charts into the REDCap database. Plan-Study-Do-Act cycles will be documented by each quality improvement team into the study database.
Data management
Sites will have individualized portals to the database with no access to other sites’ data. Participant data will be collected, used and disclosed in compliance with Alberta’s Health Information Act.34 The QIIS-II survey and interview consent and demographic data will be collected via University of Calgary Qualtrics, XM. Interview transcripts will be entered into REDCap. Access to patient identifiers will be limited to the research coordinator, data analyst and principal investigator.
Sample size
The anticipated sample size is 9500 neonates over 4 years. Power calculations account for the design effect that is determined by the number of sites and steps, the size of the clusters within each period and the intracluster correlation coefficient. 35–38 Calculations account for the relative efficiency imposed by unequal cluster sizes.39 Using historic length of stay data from 2018 and 2019, we obtained an observed intracluster correlation coefficient of 0.034 from a negative binomial model with random intercepts for sites with a corresponding design effect of 3.41. At an α level of 0.05, this provides power of greater than 99% to detect a 1-day reduction in length of stay (historic mean length of stay = 14 d). In the historic data, 81% of neonates were singletons. Sample size calculations accounting for multiple births resulted in similar power. Our sample size will ensure we have adequate power for assessments of some of the secondary outcomes that are in common with the Alberta FICare trial.10 Anticipated rates of intravenous fluid use and respiratory support will be similar to those of neonates with a gestational age of 34 weeks in the Alberta FICare data (both about 50%). Applying the same methods to power calculations for binary outcomes, we will have 80% power to detect a 10.2% relative reduction in event rates if the baseline rate is 50%, and 80% power to detect a relative reduction of 19.7% if the baseline event rate is 20%. Calculations are for a cross-sectional stepped-wedge design and were programmed using R 4.0.2, and lme4 function for generalized linear mixed effect models.40
Statistical analysis
Descriptive statistics will be used to summarize the participants’ characteristics. Negative-binomial distribution will be used to model length of stay.41 This analysis will use a generalized linear mixed model with a random intercept for the site, a fixed effect for the period, and a (0,1) indicator variable for control versus intervention periods.35,40 We will also test for a period by intervention effect to determine whether effectiveness varies over time and use random intercepts to account for multiple births. Linear mixed models will be used for continuous secondary outcomes and generalized linear mixed models for binary outcomes. As per the model for the primary outcome, these models will have random intercepts for sites and fixed effects for period and intervention. In addition, we will use QIIS-II scores of organizational culture as an effect modifier when modelling the effect of EPIQ on outcomes. Post-implementation interviews will be coded and analyzed using thematic analysis42 to identify barriers and facilitators for EPIQ implementation. We will use the CIHI Patient Cost Estimator33 to calculate the daily cost based on the Case Mix Groups for gestational age and birth weight and conduct an economic analysis using data collected in the trial and administrative data repositories in Alberta using a public health care payer perspective. Costs will include costs associated with health services as defined above over the study period. The outcome of the economic analysis is the incremental cost of the intervention compared with usual care. We will analyze the total cost variable as a dependent variable, using a linear mixed-effects regression model, to estimate the difference in expected health care cost between the 2 groups. The regression model will adjust for potential confounding variables such as gestational age, multiple births, delivery mode and urban versus regional sites.
The primary outcome will be analyzed at the end of the trial to preserve the α. Planned interim analyses of secondary outcomes that are considered quality improvement outcomes related to the care bundles will be reported for each unit every quarter using statistical process control charts. Contemporaneous change in process and outcome measures indicates effective quality improvement implementation. Benchmarking between participating NICUs will be provided annually using deidentifying codes for each NICU.
Planned subgroup analyses include NICU geographical location; gestational age subgroups, 32–34 weeks and 35–36 weeks, as admission and discharge criteria and length of stay vary between these groups; and singleton and multiple births.
Process measures and intervention fidelity
Process measures of collaborative quality improvement implementation include the number of EPIQ workshops conducted and number of participants, number of Plan-Study-Do-Act cycles completed in each unit, presentation in collaborative quality improvement meetings, and benchmarking reports. The intervention fidelity will be assessed in each unit after transition to the intervention arm by monitoring adherence to process indicators for both respiratory care and nutritional care bundles. In addition, we developed a mechanism to track quality improvement activities and operational changes in each unit as these factors may affect implementation timelines and confound the results.
Monitoring
The trial will have 3 committees. The Trial Steering Committee will oversee the trial progress and address any concern with the trial execution and management. The Trial Management Committee will be responsible for daily management of the trial, implementing EPIQ collaborative quality improvement interventions during each transition period and supporting each NICU local quality improvement team. The Data Management Committee will manage and maintain the trial database and data dictionary (Appendix 8, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1).
Substudies within the ABC-QI Trial
Evaluation of organizational culture: we will use the QIIS-II, which consists of 39 questions and will be distributed electronically to health care providers (physicians, nurses and allied health professionals) in the participating units in the first year while all units are in the control arm (Appendix 9, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1).43–45 The survey data will be analyzed after the survey closure using descriptive statistics. The results will help the investigators understand the organizational culture, the status of quality improvement implementation, and differences between units in order to support quality improvement team building and implementation, and might help us interpret some of the differences in outcomes.
Evaluation of collaborative quality improvement implementation: this substudy aims to understand the barriers and facilitators in implementing EPIQ collaborative quality improvement interventions. Methods: semistructured interviews with a purposively selected sample of 4 health care providers from different disciplines in each NICU, who are not part of the quality improvement team. The interview questions (Appendix 10, available at www.cmajopen.ca/content/11/3/E397/suppl/DC1) are based on the Consolidated Framework for Implementation Research and spread over 5 domains: intervention characteristics, outer setting, inner setting, individual characteristics and process.46 The interview data will be analyzed using qualitative methodology at the end of the ABC-QI Trial.
Ethics approval
The study has been approved by the University of Calgary Conjoint Health Research Ethics Board (REB21-1336), University of Alberta Health Research Ethics Board, Alberta Health Services and Covenant Health.
Interpretation
The ABC-QI study aims to provide real-world evidence of the benefit of collaborative quality improvement strategies in moderate and late preterm neonates. The study will use quality improvement tools as an intervention, which carries the potential for implementation of better practices and providing directions for future research in moderate and late preterm neonates. Furthermore, knowledge sharing will assist in collaboration and build collective knowledge for future research. We will disseminate the results to health care professionals, parent advisory groups, operational leaders, policy-makers, government agencies and other knowledge users through presentation in national and international conferences, media sources and publication in relevant peer-reviewed journals using the Consolidated Standards of Reporting Trials extension for stepped-wedge cluster randomized trial.47
Limitations
EPIQ methodology aims for change in practice and culture. These changes follow the change cycle described by Prochaska and DiClemente, in which the last stage is relapse.48 We will mitigate this risk through ongoing engagement, quarterly benchmarking reports and the continuous capability of each unit to access their data. The study aims to implement 2 care bundles within 12 months of transition to intervention. Barriers due to operational changes or resourcing may limit the ability of the units to implement quality improvement initiatives.
Conclusion
The ABC-QI trial aims to evaluate the impact of collaborative quality improvement strategies in moderate and late preterm neonates on clinical outcomes, length of stay and cost to health care system on a wide scale. Our goal is to address the existing research gaps in moderate and late preterm neonates and provide detailed population-based data that can be used for future research, benchmarking and quality improvement in the population of moderate and late preterm neonates across Canada and worldwide.
Acknowledgements
The authors thank the site medical and operational leads, Maternal, Newborn, Child and Youth Strategic Clinical Network, and Alberta SPOR SUPPORT Unit for their support of the project. The authors also thank the Canadian Neonatal Network for providing and sharing its data dictionary, the Canadian Neonatal Foundation for the EPIQ materials and the University of Berkeley for sharing the Quality Improvement Implementation Survey.
Footnotes
Competing interests: Khalid Aziz is a paid facilitator of Evidence-based Practice for Improving Quality (EPIQ) workshops delivered by the University of Alberta and is a volunteer board member of the Canadian Neonatal Foundation. Dr. Aziz reports grants from McGill University for teaching EPIQ for the School of Business. He has received payment from the Canadian Medical Protective Association for legal advice unrelated to this project and was funded by Canadian Institutes of Health Research (CIHR) to attend QI meetings. Charlotte Foulston reports clinical fee-for-service payments from Alberta Health as an Alberta Health Services pediatrician working in the Medicine Hat Regional Hospital neonatal intensive care unit since 2010 (no payment directly related to this manuscript) and payment from Alberta Health Services as clinical pediatrics department lead in Medicine Hat from 2011 to Mar. 1, 2022 (no payment related to this manuscript). Dr. Foulston is a Maternal, Newborn, Child and Youth (MNCY) Strategic Clinical Network (SCN) core committee member in Alberta and has received hourly payment for attending meetings. MNCY SCN has had presentations regarding this study and is aware and supportive of the work; Dr. Foulston has not received payment related to this manuscript. Dr. Foulston is a member of the MNCY SCN Neonatal Intensive Care Unit Steering Committee; she has received a stipend for attending meetings and has not received payment related to this study. Gabrielle Zimmermann and Yazid Al Hamarneh work for the Alberta SPOR [Strategy for Patient-Oriented Research] SUPPORT [Support for People and Patient-Oriented Research and Trials] Unit, which is funded in part by CIHR. Leonora Hendson reports being a member of the Early Hearing Detection and Intervention Program Advisory Committee and the Provincial Perinatal Quality Assurance Committee (neither related to this manuscript or project). No other competing interests were declared.
This article has been peer reviewed.
Contributors: Ayman Abou Mehrem, Jennifer Toye, Khalid Aziz, Nalini Singhal, Prakesh Shah, Karen Benzies, Belal Alshaikh, David Johnson, Peter Faris, Yazid Al Hamarneh, Gabrielle Zimmermann and Deborah McNeil developed the initial concept and design of the trial. All authors reviewed and approved the design of the trial. Ayman Abou Mehrem, Jennifer Toye, Khalid Aziz, Nalini Singhal, Prakesh Shah, Karen Benzies, Belal Alshaikh, David Johnson, Amuchou Soraisham, Peter Faris, Yazid Al Hamarneh, Gabrielle Zimmermann, Charlotte Foulston, Karen Foss, Deborah McNeil and Christine Johns drafted sections of the grant application. Ayman Abou Mehrem, Dana Price and Khalid Aziz drafted the manuscript. All authors reviewed and approved the initial draft and final version of the manuscript and agreed to be accountable for all aspects of the work.
Funding: The ABC-QI Trial Investigators are supported by grant no. 178288 from the Canadian Institutes of Health Research.
Data sharing: After study completion, the deidentified, archived data will be transmitted to and stored at the Federated Research Data Repository, for use by other qualified researchers.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/3/E397/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors