Abstract
Background: Choosing Wisely is a high-profile campaign seeking to reduce the use of low-value care. We investigated the impact of a Choosing Wisely Canada recommendation against using a combination of angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) for the management of hypertension, heart failure or diabetic nephropathy on population-level use of these medications in British Columbia, Canada.
Methods: We identified all people (any age) who were continuously registered with BC’s Medical Service Plan between 2010 and 2017 with the targeted conditions. Using prescription claims data and an interrupted time-series analysis, we estimated the number of people on combination therapy per month, the proportion of days covered (PDC) by combination therapy per month and proportion of all combination prescriptions started per month in the 2 years before and after the introduction of the recommendation on Oct. 29, 2014.
Results: Of 1 104 593 people (mean age 65 yr, standard deviation 16 yr) in our study cohort, 4.6% were exposed to combination therapy, largely prescribed by family physicians (84%). The number of people on combination therapy and the PDC were declining before the recommendation, but the proportion of combination prescriptions started in the 2 years before the recommendation was increasing. After the recommendation, we observed no statistically significant changes in any outcome. The pre-existing downward trend of the monthly number of people decelerated (16.8, 95% confidence interval [CI] 14.0 to 19.5) and the proportion of prescriptions started increased (0.13%, 95% CI 0.08% to 0.18%).
Interpretation: The Choosing Wisely Canada recommendation against using a combination of ACE inhibitors and ARBs was not associated with reduced combination therapy use in the targeted conditions. The observed pre-existing declines in this practice questions the process of selecting recommendations, and the optimal implementation and value of Choosing Wisely campaigns without other reinforcing interventions.
Choosing Wisely is an international campaign that seeks to reduce waste in health systems by reducing the use of low-value care, namely medical treatments, services and procedures that offer no or little benefit.1 In Canada, more than 300 recommendations have been released since 2014, with almost 70 clinician societies participating in their development.2 Each medical specialty develops specialty-relevant, evidence-based lists that recommend unnecessary tests and treatments to avoid. These recommendations are available to physicians (e.g., through a mobile app, on the campaign website) and to patients (e.g., via posters displayed in physician offices). Implementation of each recommendation is specific to the context. Several provincial-and territorial-specific campaigns have been developed to prioritize and accelerate the adoption of regionally relevant recommendations from the national campaign.3
Several Choosing Wisely recommendations target low-value pharmaceutical use, but the evidence on whether Choosing Wisely influences prescribing is not equivocal.4,5 On Oct. 29, 2014, the Canadian Society of Nephrology recommended against the use of combination angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) (hereafter, combination therapy) for the treatment of hypertension, diabetic nephropathy or heart failure.6 This recommendation was made on the basis of rigorous clinical evidence that showed an associated increased risk of symptomatic hypotension, acute renal failure, hyperkalemia and death, and was supported by the 2014 Eighth Joint National Committee and the 2014 Canadian Hypertension Education program guideline.7–9 A previous population-level study in Alberta in 2011 found that 5.4% of new users of ACE inhibitors or ARBs received combination therapy;10 however, the extent of use in other provinces before and after the recommendation from Choosing Wisely Canada is not known. Given the strength of evidence supporting this recommendation, and the lack of uncertainty in its clinical application, we hypothesized that, if Choosing Wisely campaigns were effective, this is an instance where a change in prescribing should be observable. We sought to evaluate the impact of introducing the recommendation from Choosing Wisely Canada on changes in population-level use of combination therapy for the treatment of hypertension, diabetic nephropathy or heart failure in British Columbia, Canada.
Methods
Study design
We used a quasiexperimental interrupted time-series analysis11 to determine the impact of the Choosing Wisely Canada recommendation of “Do not prescribe angiotensin converting enzyme (ACE) inhibitors in combination with angiotensin receptor blockers (ARBs) for the treatment of hypertension, diabetic nephropathy and heart failure,” which was publicly released in Canada on Oct. 29, 2014.6 Interrupted time-series analyses are widely used in health services research because, unlike most other longitudinal observational research designs, pre-existing secular trends in outcomes are controlled for so that causal effects of an intervention can be estimated.11–14 We report our study with reference to the Reporting of Studies Conducted Using Observational Routinely-collected Health Data (RECORD) checklist.15
Setting
We conducted our study in BC, which has almost 5 million people at an average age of 42.3 years and is the third-most populous Canadian province.16,17 Residents of BC are offered public coverage for medically necessary services via the Medical Services Plan (MSP). Although there is no similar public coverage for prescription medicines, every prescription supplied from a community pharmacy in BC is recorded in a province-wide database (BC PharmaNet), which makes it possible to track drug use and expenditure over time.18
Study population
We included all people with diagnoses of hypertension, heart failure or diabetic nephropathy. Patients could have more than 1 diagnosis. We excluded people who ceased enrolment in MSP for reasons other than death. In addition, we excluded people who were in receipt of drug benefits through the Federal government (e.g., First Nations, military) because data about their drug use and expenditure were not captured in PharmaNet.
Data sources
We used 6 population-based data systems on use of health services in the province between 2010 and 2017 (Appendix 1, Table S1, available at www.cmajopen.ca/content/10/4/E1059/suppl/DC1). We obtained information on fee-for-service physician consultations and expenditures through the MSP billings data,19 and hospital admission information from the Discharge Abstract Database.20 These were used to define our population using International Classification of Diseases, 9th Revision (ICD-9) and 10th Revision (ICD-10) codes, which have been previously used in the literature for the target conditions.21,22 We included people with 1 code recorded in the Discharge Abstract Database or 2 of the same codes recorded in the MSP within 2 years. For diabetic nephropathy, we included all people with diabetes and any kidney-related complication.
We used the BC PharmaNet database to track drug use.23 We obtained demographic information about the population from the MSP registry file and Vital Statistics Mortality data.24,25 We obtained prescriber specialty information from the College of Physicians and Surgeons of BC data set,26 which was linked via a unique practitioner identifier. These data sets were linked using a unique patient health number by Population Data BC.
Outcomes
We measured the number of people on combination therapy per month, the mean proportion of days covered (PDC) for combination therapy per month and the proportion of combination therapy prescriptions started per month. We defined combination treatment as exposure to both an ACE inhibitor and an ARB at the same time, as indicated by dispensing data. To determine combination treatment, we identified all prescriptions containing an ACE inhibitor or ARB using the Anatomic Therapeutic Classification codes C09A-D.27 For each individual, we created a matrix that indicated if an ACE inhibitor or ARB was dispensed. We used a gap in treatment of more than 90 days to indicate treatment cessation. We created a variable to indicate in each month when combination therapy was started or stopped. For each instance of combination therapy, we recorded the specialty of the physician (i.e., nephrologist, family physician or other) who prescribed the second agent in the combination.
To calculate the PDC for combination treatment, we first determined the PDC for any ACE inhibitor or ARB-containing medicine and then determined the PDC when both ACE inhibitors and ARBs were available per month in the overall cohort. The PDC is a ratio between 0 and 1 of the number of days in a period when a medication is available divided by the number of days in the period.28 The PDC is endorsed by the National Quality Forum for measuring health care quality.29 A decrease in PDC represents reduced medication use. We expressed the PDC as a percentage and inferred reduced PDC as reduced exposure and, therefore, reduced use of combination therapy.
Statistical analysis
In our analyses, we estimated the change in the immediate level and the trend of each outcome 24 months before and after the public release of the recommendation from Choosing Wisely Canada. We weighted our analysis by population size to adequately account for deaths in our cohort, and proximity of time to implementation of the recommendation, with the biggest weight given to the first implementation observation, and more weight given to time points that were closer to the implementation date. We weighted by proximity to implementation time because observations that were closer to the implementation time were likely to contain relatively more information (i.e., they are more informative about the underlying relationship between X and Y). Weights were specified to assume that the variance of the error term was proportional to the population of the cohort. Our evaluation of weight selection consisted of a comparative analysis involving our modelling approach without weight, with population size weight, and with population size weight and time proximity weight.30 From this comparative analysis, the combined population size weight and the proximity time weight produced the most precise estimates with narrower confidence intervals (CIs), which indicated the increased statistical power; thus, we chose this as our final weight. This weighting accounts for the dynamic change of cohort size and time proximity change over time. We used the generalized least squares regression and included appropriate factors to account for the autocorrelation in the residuals (i.e., autoregressive process of order 1).
As the Choosing Wisely Canada recommendation was endorsed by the Canadian Society of Nephrology, we performed a secondary stratified analysis by the specialty of the physician (i.e., nephrologist, family physician, other) who prescribed the second agent in the combination. We also performed stratified analysis by patient age (≤ 65 yr, > 65 yr) and sex.
Ethics approval
The study was approved by the University of British Columbia Behavioural Research Ethics Board (H16-02087).
Results
Our population included 1 104 593 people with a diagnosis of hypertension, heart failure or diabetic nephropathy (Appendix 1, Figure S1). Our sample was 50.1% female and 51.3% were aged 65 years or older (Table 1) at the time of the Choosing Wisely Canada recommendation. Most people (77.6%) in our sample had a diagnosis of hypertension, predominantly essential hypertension (77.3%), while only 13.0% had heart failure.
Characteristics of population with hypertension, diabetic nephropathy or heart failure, overall and among those dispensed combination angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs)*
Proportion of combination use
Just under 5% of our population received combination therapy at some point before or after the Choosing Wisely Canada recommendation. Although the demographic characteristics of people on combination therapy were similar to those of the overall population, we noted lower proportions of people with diabetic nephropathy and younger people on combination therapy. Most of the combination therapy in our sample was prescribed by family physicians (n = 86 873 prescriptions, 82.7%), as opposed to nephrologists (n = 4481, 4.3%) or other specialists (n = 13 709, 13.0%).
Number of people per month on combination treatment
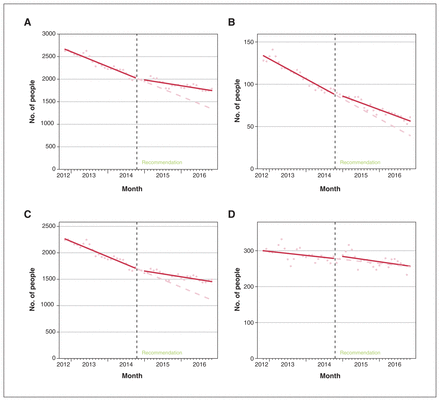
Figure 1A displays the results from the interrupted time-series analysis for the number of people per month on combination treatment. Before the recommendation from Choosing Wisely Canada, the number of people on combination treatment per month was declining (−27.5, 95% CI −29.5 to −25.5). After the recommendation, we did not see a statistically significant change in his number (18.5, 95% CI −21.9 to 58.8). Counter to expectations, we found an attenuation of the pre-existing declining trend of the number of people on combination treatment per month, with a statistically significant increase of 16.8 people per month (95% CI 14.0 to 19.5).
(A) Interrupted time-series results of the number of people on combination therapy of angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) 24 months before and after the Choosing Wisely Canada recommendation. (B) Interrupted time-series results for the number of people on combination ACE inhibitors and ARBs per month prescribed by nephrologists, (C) family physicians and (D) other physicians (i.e., specialties other than nephrologists or family physicians).
When stratified by prescriber specialty, we saw similar trends (Figure 1B–D). The number of people per month on combination therapy was declining in the 24 months before the Choosing Wisely Canada recommendation for nephrologists (−2.0, 95% CI −2.3 to −1.6), family physicians (−24.1, 95% CI −27.0 to −21.1) and other physicians (−0.9, 95% CI −1.8 to −0.1). After the recommendation, there was no significant change in the levels. However, there was a statistically significant attenuation of the pre-existing declining trend for nephrologists (0.6, 95% CI 0.1 to 1.1) and family physicians (15.0, 95% CI 10.7 to 19.2), but not for other specialties (−0.3, 95% CI −1.5 to 0.9).
Proportion of days covered for combination therapy
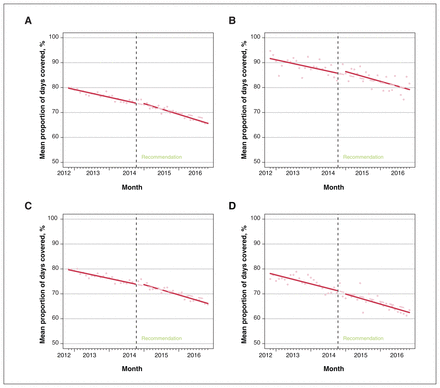
Figure 2A displays the results from the interrupted time-series analysis for mean PDC for combination treatment per month. In the 2 years before the Choosing Wisely Canada recommendation, the mean PDC was decreasing (−0.26%, 95% CI −0.29% to −0.22%). After the recommendation, we did not see a statistically significant change in the mean PDC (0.63%, 95% CI −0.05% to 1.32%), but did observe a small, statistically significant acceleration of the pre-existing trend by 0.10% per month (95% CI −0.15% to −0.06%).
(A) Interrupted time-series results for the mean proportion of days covered (PDC, %) for combination therapy of angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) per month in the 24 months before and after the Choosing Wisely Canada recommendation. (B) Interrupted time-series results for the mean PDC for combination therapy of ACE inhibitors and ARBs per month for nephrologists, (C) family physicians and (D) other physicians (i.e., specialties other than nephrologists or family physicians).
When stratified by prescriber specialty, we saw similar trends (Figure 2B–D). The mean PDC for combination therapy was declining in the 24 months before the Choosing Wisely Canada recommendation for nephrologists (−0.25%, 95% CI −0.41 to −0.10), family physicians (−0.25%, 95% CI −0.29 to −0.21) and other physicians (−0.30%, 95% CI −0.40 to −0.19). After the recommendation, there was no significant change in the levels. However, we observed a statistically significant increase in the pre-existing declining trend for family physicians (−0.09%, 95% CI −0.15 to −0.04), but not for nephrologists (−0.08%, 95% CI −0.31 to 0.15) or other specialties (−0.04%, 95% CI −0.19 to 0.11).
Proportion of combination therapy prescriptions started per month
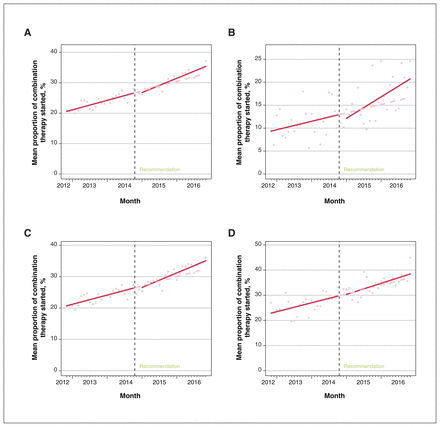
Figure 3A displays the results from the interrupted time-series analysis for the proportion of all combination therapy prescriptions that were started per month. Before the recommendation from Choosing Wisely Canada, the proportion of combination treatment prescriptions started was increasing by 0.26% per month (95% CI 0.22% to 0.30%). After the recommendation, we saw a trend toward a reduction in the immediate proportion of new prescriptions that was not statistically significant (−0.69%, 95%CI −1.38% to 0.01%), and, counter to expectations, we saw a statistically significant increase in the pre-existing trend (0.13%, 95% CI 0.08 to 0.18).
(A) Interrupted-time series results for the proportion of all prescriptions of combination therapy of angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) started per month in the 24 months before and after the Choosing Wisely Canada recommendation. (B) Interrupted-time series results for the proportion of prescriptions of combination therapy of ACE inhibitors and ARBs started per month for nephrologists, (C) family physicians and (D) other physicians (i.e., specialties other than nephrologists or family physicians).
We saw similar results when this outcome was stratified by prescriber specialty (nephrologists 0.15%, 95% CI 0.07% to 0.24%; family physicians 0.24%, 95% CI 0.20% to 0.29%; other 0.29%, 95% CI 0.11% to 0.47%) (Figure 3B–D), with the trend toward a statistically significant reduction in the immediate level evident only with nephrologists (−1.40%, 95% CI −3.03% to 0.17%) and the increase in the pre-existing trend not statistically significant for physicians who were not family physicians or nephrologists (0.08%, 95% CI −0.19% to 0.34%).
Stratified analyses by sex and age (≥ 65 v. < 65 yr) were also examined, but did not show any differences except for a statistically significant increase in the level of mean PDC for females immediately after the Choosing Wisely Canada recommendation (1.56%, 95% CI 0.72% to 2.39%) (not shown, available upon request).
Interpretation
In this study, we evaluated the impact of the recommendation from Choosing Wisely Canada that sought to reduce combination ACE inhibitor and ARB use for the treatment of hypertension, heart failure and diabetic nephropathy. Despite this recommendation being supported by strong clinical evidence with clear clinical application,7,8 in our rigorous population-level analysis, we did not find unequivocal evidence to support the effectiveness of Choosing Wisely on reducing this low-value pharmaceutical practice. Although the rate of decline of exposure to combination therapy after the recommendation was attenuated, counter to expectations, we found the declining rate of people on combination therapy decelerated in the 2-year period after the recommendation, along with the proportion of combination prescriptions started in this period.
Using rigorous analytical methods that can account for secular trends and pre-existing patterns of use, we found that this recommendation from Choosing Wisely Canada had no immediate impact on an infrequently used, low-value pharmaceutical practice, and attenuated some of the reductions already occurring over time. We also found that the extent of combination therapy was comparable to other provinces,10 but represented less than 5% of our population, and that most prescribing was performed by family physicians, rather than the society that endorsed the recommendation. These findings reinforce previous concerns raised about the selection of low-value practices,3,31,32 and supports calls to improve methods of identifying high-priority clinical targets to fulfil the promise of Choosing Wisely campaigns.33
Our study adds to the existing body of evidence that suggests that Choosing Wisely recommendations alone may have little impact on changing the use of the targeted low-value pharmaceuticals. 4,5 It has been argued that the lack of measurable impact of Choosing Wisely is because additional interventions are needed to permit wider and more sustained implementation. 33,34 This may be particularly relevant in BC where the Choosing Wisely Canada campaign received less investment than other Canadian provinces, albeit anecdotally.31 However, the inability of this Choosing Wisely Canada recommendation to change physician behaviour is not surprising, given the large body of evidence that questions the impact of passive, generalized, physician-targeted information provision campaigns to change physician behaviour, including prescribing practices.35–38 Behavioural nudge approaches and other theory-based interventions may improve the implementation of Choosing Wisely.33,39 Considering the large investments already made in the Choosing Wisely campaign, however, the incremental costs and benefits of a modified Choosing Wisely approach, compared with its current format, should be further evaluated, as should the economic viability of this approach, compared with other strategies of decreasing low-value prescribing practices.
Limitations
As Choosing Wisely Canada was implemented nationally at a fixed date, there was not an appropriate control group that could be used in our analysis. Further, we did not have the exact clinical context and there may be individuals for whom combination therapy may be appropriate (e.g., those with severely uncontrolled heart failure alongside strict monitoring) but who we were not able to identify from administrative data nor perform sensitivity analyses. However, we expect this would have been a very small proportion of our population,40 and given that the Choosing Wisely Canada intervention does not affect an individuals’ clinical criteria, we would not expect the proportion of such cases over time to have changed because of Choosing Wisely Canada, which would otherwise threaten the validity of the interrupted time-series analysis. It is also possible that the number of people on combination treatment may have been overestimated given that our 90-day definition of cessation could not exclude people who may have reasonably switched from 1 class to another (e.g., owing to adverse effects). There may also have been other interventions to reduce combination therapy prescribing (e.g., physician detailing) that were implemented at the same time as the Choosing Wisely Canada campaign of which we were unaware, which could affect our effect estimates. However, as the pivotal evidence for this recommendation and associated updates to clinical guidelines occurred many years before the recommendation, we expect this to be unlikely. It is unclear if the Choosing Wisely Canada campaign was as active in BC as other provinces, so these results may not fully extrapolate to other settings.
Conclusion
The release of the recommendation from Choosing Wisely Canada against the concomitant use of ACE inhibitors and ARBs for the management of hypertension, heart failure or diabetic nephropathy in BC did not reduce combination therapy alone. Our findings reinforce the limited effectiveness of passive strategies of information provision to improve medicine use. Future consideration of coupling Choosing Wisely with other established interventions of behaviour change, with rigorous evaluation of its effects, is suggested. The observed pre-existing declines in the use of this low-value practice also calls into question the process of selecting recommendations.
Footnotes
Competing interests: Michael Law reports consulting fees from Health Canada and the Hospital Employees’ Union, and has provided expert witness testimony for the Attorney General of Canada and the Federation of Post-Secondary Educators. No other competing interests were declared.
This article has been peer reviewed.
Contributors: All of the authors contributed to the conception and design of the work, and the acquisition, analysis and interpretation of data. All of the authors drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This analysis was funded by a Foundation Scheme Grant from the Canadian Institutes of Health Research (FDN-148412, Principal Investigator Michael Law). Michael Law received salary support through a Canada Research Chair in Access to Medicines and a Michael Smith Foundation for Health Research Scholar Award. Tracey-Lea Laba received salary support through a National Health and Medical Research Centre Early Career Fellowship (APP 1110230).
Disclaimer: Data for this study were obtained via Population Data BC, including data from the BC PharmaNet system. All inferences, opinions and conclusions drawn in this publication are those of the authors and do not reflect the opinions or policies of the Data Steward(s).
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/4/E1059/suppl/DC1
Data sharing: The data that support the findings of this study are available from Population Data BC (https://www.popdata.bc.ca/) but restrictions apply to the availability of these data, which were used for the current study under Data Stewards’ approval and research agreements with data stewards, and so are not publicly available. Data are, however, available from Population Data BC upon reasonable request and with permission of the Data Stewards.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use) and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors