Abstract
Background: Psychological distress following a cancer diagnosis potentially increases the risk of intentional, nonfatal self-injury. The purpose of this work is to evaluate and compare rates of nonfatal self-injury among individuals in Ontario diagnosed with cancer against matched controls with no history of cancer.
Methods: Adults in Ontario diagnosed with cancer from 2007 to 2019 were matched to 2 controls with no history of cancer, based on age and sex. We calculated the absolute and relative difference in rates of nonfatal self-injury in the 5 years before and after the index date (date of cancer diagnosis and dummy date for controls). We used crude difference-in-differences methods and adjusted Poisson regression-based analyses to examine whether the change in rates of nonfatal self-injury before and after index differed between cancer patients and controls.
Results: The cohort included 803 740 people with cancer and 1 607 480 matched controls. In the first year after diagnosis, individuals with cancer had a 1.17-fold increase in rates of nonfatal self-injury (95% confidence interval [CI] 1.03–1.33) compared with matched controls, after accounting for pre-existing differences in rates of nonfatal self-injury and other clinical characteristics between the groups. Rates of nonfatal self-injury remained elevated in the cancer group by 1.07-fold for up to 5 years after diagnosis (95% CI 0.95–1.21).
Interpretation: In this study, incidence of nonfatal self-injury was higher among individuals diagnosed with cancer, with the greatest impact observed in the first year after diagnosis. This work highlights the need for robust and accessible psychosocial oncology programs to support mental health along the cancer journey.
After a cancer diagnosis, individuals are often faced with real risks of death and disability.1–3 In times of prolonged stress, individuals are particularly susceptible to depressive symptoms or severe manifestations of mental illness, including self-injury or suicide.4,5 In Canada, rates of death by suicide are 1.3 times higher for individuals with cancer than for the general population.5 In other countries, suicide rates have been reported to be up to 4.4 times higher than in the general population.6–8 Although a substantial amount of work has focused on suicide risk among those diagnosed with cancer,9 broader manifestations of mental illness, such as nonfatal self-injury, have not been as well studied. We recently established that 3 in 1000 patients will experience a nonfatal self-injury event after their cancer diagnosis.10 However, data are lacking on how this compares with the general population. Identifying whether individuals diagnosed with cancer are at an increased risk for nonfatal self-injury is important in devising and funding supportive care programs for patients with cancer.
Given this, we sought to compare the rate of nonfatal self-injury among individuals in Ontario diagnosed with cancer against matched controls with no history of cancer.
Methods
We conducted a matched cohort study examined under a difference-in-differences framework using population-based routinely collected administrative data. All data sets used in this study were linked using unique encoded identifiers and analyzed at ICES. This study was reported according to the Reporting of Studies Conducted Using Observational Routinely-collected Health Data (RECORD) statement.11
Data sources
The Ontario Cancer Registry (OCR) is a provincial database on individuals diagnosed with cancer other than basal cell carcinoma and squamous cell carcinoma of the skin. The OCR is estimated to capture more than 95% of all diagnoses in Ontario.12 The Registered Persons Database contains demographic information for all individuals who are eligible for the Ontario Health Insurance Plan (OHIP).13 The Discharge Abstract Database contains patient-level data, including clinical data, demographic data and administrative data for acute, rehab, chronic and day surgery institutions in Ontario.14 The National Ambulatory Care Reporting System captures information on patient visits to hospitals and community-based ambulatory care. The Ontario Mental Health Reporting System contains information from participating hospitals in Ontario that report on patients’ psychiatric diagnoses or usage of mental health services. The Ontario Marginalization Index is a geographically based index that quantifies the degree of marginalization across Ontario based on Canadian census data.15
Study cohort
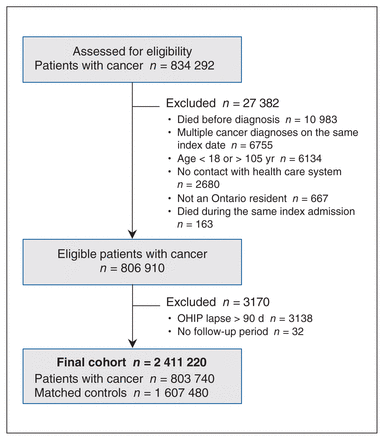
Individuals aged 18 years or older diagnosed with cancer between Jan. 1, 2007, and Mar. 31, 2019, as identified in the OCR using International Classification of Diseases for Oncology, 3rd edition codes,16 were selected for potential inclusion in the study (Appendix 1, available at www.cmajopen.ca/content/11/2/E291/suppl/DC1). Individuals were subsequently excluded if they had more than 1 cancer diagnosis on the same day, if their date of last contact was missing, if their death date preceded their cancer diagnosis date (indicating an entry error), if they were ineligible for OHIP on the date of diagnosis, or if their OHIP eligibility lapsed for a period of greater than 90 days in the 5 years before diagnosis.
Two control individuals without a cancer history were selected for each individual with cancer, based on a hard match of age (exact birth year) and sex. For individuals with cancer, their index date was the date of cancer diagnosis. Individuals in the control group were assigned the same index date as the matched cancer patient. We collected information for up to 5 years before the index date. Individuals were then followed from the index date until their date of death, the date that they lost OHIP eligibility, or Mar. 31, 2020, whichever occurred first.
Covariates
All covariates were measured at the index date. Age and sex were categorical variables. Rurality was dichotomized as rural or urban using the Rurality Index for Ontario.17 Material deprivation, a measure of socioeconomic status, was categorized into quintiles with the fifth quintile representing the highest level of deprivation (most deprived).15 Prior usage of mental health services in the 5 years before the index date was categorized as no mental health service use, inpatient psychiatric care, outpatient psychiatric care or other mental health usage, as previously described.18,19 We captured the presence of comorbidities in the 2 years before the index date using a modified version of the Elixhauser Comorbidity Index that excluded cancer diagnoses and was dichotomized as low (0–3) or high (≥ 4).20,21 For individuals with cancer, cancer stage, cancer type and year of diagnosis (grouped by 2-year intervals) were reported. We reported cancer stage as per the American Joint Committee on Cancer Staging Manual, 7th edition22 (Appendix 2, available at www.cmajopen.ca/content/11/2/E291/suppl/DC1).
Outcome
The outcome of interest was the rate of nonfatal self-injury. Based on prior work, nonfatal self-injury incidence was defined as an emergency department visit for self-injury (including physical injury or self-poisoning) of intentional (International Statistical Classification of Diseases and Related Health Problems, 10th Revision, Canada [ICD-10-CA] codes X61–X84) or undetermined intent (ICD-10-CA codes Y10–Y19, Y28).23–26
Statistical analysis
Baseline characteristics We compared the distributions of baseline characteristics between cancer patients and corresponding matched controls. Between-group comparisons of proportions were performed using standardized differences.27 We defined a significant imbalance as a weighted standardized difference of 0.10 or greater.27
Selection of pre- and postindex periods
We employed a crude difference-in-differences analysis comparing absolute rates of nonfatal self-injury and an adjusted Poisson regression–based analysis. For both approaches, the rate of nonfatal self-injury in the cancer group and the control group were calculated in the first year after the index date (year 0–1) and compared with the rate in the 5 years before the index date. Rates in years 0–5 and 1–5 were also calculated and compared with preindex rates of nonfatal self-injury.
The recorded date of diagnosis for an individual is not necessarily the date that they become aware of their cancer diagnosis or the date that they begin to experience symptoms associated with their diagnosis. In fact, individuals may enter the health care system with cancer-related symptoms up to 1 year before they receive a diagnosis; this is referred to as the peridiagnostic period.28 For this reason, there is the potential that any nonfatal self-injury events that occur in the year before diagnosis may not be reflective of the baseline rate of self-injury, but may instead be related to the cancer diagnosis. To ensure that we captured a true preindex rate of nonfatal self-injury that was unaffected by the cancer experience, we repeated our analyses excluding the peridiagnostic period, allowing for a 6-month peridiagnostic period, by excluding all nonfatal self-injury events and follow-up time in the 6 months immediately preceding the index date. This was repeated allowing for a 12-month peridiagnostic period.
Crude difference-in-differences analysis
The rate of self-injury was calculated as the number of events in a given period, divided by the sum of the person-years in that period to account for differences in follow-up time. For the crude analysis, the difference in rates (cancer rate minus control rate) and relative rates (cancer rate divided by control rate) were calculated. The crude difference-in-differences was then calculated by subtracting the difference obtained in the preindex period from the difference obtained in the postindex period. The ratio of relative rates was calculated by dividing the relative rate of nonfatal self-injury postindex by the relative rate preindex.
Poisson regression–based difference-in-differences analysis
For the regression-based analysis, we implemented a Poisson regression model using generalized estimating equations to account for the matched design. The models used the natural logarithm of each individual’s follow-up time as the offset. The unadjusted analysis modelled the outcome rate and included 3 necessary covariates: exposure (cancer or control), period (pre- or postindex), and an interaction between exposure and period. The adjusted model included any measured covariates that showed imbalance between the cancer and control groups at index. All analyses were performed using SAS Enterprise Guide 7.1 (SAS Institute) and R Studio 12.1 (R Foundation).
Ethics approval
Studies conducted at ICES using administrative data fall under section 45 of Ontario’s Personal Health Information Protection Act and do not require research ethics board approval.
Results
The final study cohort included 803 740 individuals with cancer and 1 607 480 matched controls (Figure 1, Table 1). Over the entire study period, there were 6708 nonfatal self-injury events in the cancer group and 13 070 in the control group. In the preindex period, the mean follow-up time in the cancer and control groups were 1770 days and 1763 days, respectively. In the postindex period, the mean follow-up times were 1596 days in the cancer group and 1949 days in the control group (Appendix 3, available at www.cmajopen.ca/content/11/2/E291/suppl/DC1).
Cohort creation. Note: OHIP = Ontario Health Insurance Plan.
Distributions of baseline characteristics among patients with cancer and cancer-free controls
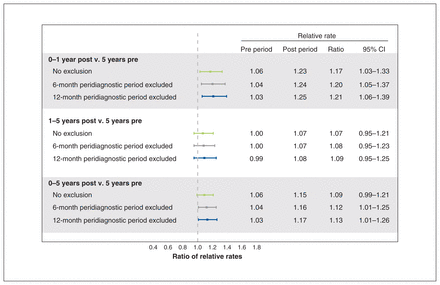
In the 5 years before the index date, there were 9.37 (95% confidence interval [CI] 9.07–9.68) events per 10 000 person-years of follow-up time among individuals with cancer and 8.64 (95% CI 8.44–8.85) events per 10 000 person-years among controls (relative rate 1.08, 95% CI 1.04–1.13). In years 0–1 after the index date, there were 10.40 (95% CI 9.66–11.19) events per 10 000 person-years among individuals with cancer and 8.24 (95% CI 7.80–8.71) events per 10 000 person-years among controls (relative rate 1.26, 95% CI 1.15–1.38) (Table 2). The adjusted ratio of relative rates obtained from the regression model was 1.17 (95% CI 1.03–1.33), indicating that after accounting for pre-existing differences in rates of nonfatal self-injury between the 2 groups, rates in the cancer group remained 1.17 times higher after diagnosis compared with the control group (Figure 2). When the analysis was repeated excluding the peridiagnostic period (assuming a 6-month peridiagnostic period) we observed a 1.20-fold (95% CI 1.05–1.37) increase in nonfatal self-injury in the cancer group compared with the control group. Assuming a 12-month peridiagnostic period, we observed a 1.21-fold (95% CI 1.06–1.39) increase. The ratio of relative rates of nonfatal self-injury between the cancer and control group in years 1–5 after index was lower compared with years 0–1 (relative rate 1.07, 95% CI 0.95–1.21) (Figure 2).
Difference-in-differences calculation of nonfatal self-injury in the first 5 years after diagnosis
Adjusted ratio of relative rates of nonfatal self-injury in patients with cancer compared with controls. Estimates have been adjusted for Elixhauser Comorbidity Index score (dichotomized as low [0–3] or high [≥ 4]).
Interpretation
In this population-based difference-in-differences study, we compared rates of nonfatal self-injury between individuals with and without cancer who were matched by age and sex. The relative increase in nonfatal self-injury among individuals with cancer was 1.17 times higher in the year after diagnosis, compared with controls, over the same period. Rates of self-injury were not significantly elevated after 1 year. When we excluded the peridiagnostic period from the analysis, we observed a greater difference-in-differences in rates of nonfatal self-injury between the 2 groups. The peridiagnostic period is a noted time of distress,29 creating the potential for increased nonfatal self-injury. Including these events likely artificially inflates the preindex rates of nonfatal self-injury in the cancer group, thereby decreasing the observed difference-in-differences estimate.
Although nonfatal self-injury is recognized as a crucial repercussion of critical illness or other traumatic events, such as major burns, it has not been examined in patients with cancer.30,31 The literature on critical illness identified different sets of risk factors for self-injury in different clinical groups.31 These results suggest that risk factors for self-injury may be context specific, supporting the need for cancer-specific research to identify risk factors for self-injury among individuals with cancer. The increased rate of nonfatal self-injury after cancer diagnosis observed in this study, particularly in the first year after diagnosis, mirrors trends in suicide after cancer diagnosis.5–7 Though this study assessed rates of nonfatal self-injury, which may include self-injury with suicidal intent as well as self-injury without suicidal intent, these 2 behaviours have overlapping risk factors, such as depression, anxiety and hopelessness, related to cancer-associated poor mental health.32–34 This work adds to the existing literature on mental illness among individuals with cancer by reporting on nonfatal self-injury as a target outcome for potentially severe manifestations of poor mental health.
Recognizing that individuals with cancer have an increased risk for self-injury compared with the general population confirms that patients with cancer require additional support and resources to manage poor mental health throughout the continuum of their care. Our prior work has identified that younger age, certain cancer subsites (including head and neck cancers), history of severe psychiatric illness and prior self-injury were independently associated with risk of nonfatal self-injury.10 Furthermore, these exposures act synergistically, placing young adults with a prior mental health history at the greatest risk of nonfatal self-injury. Such high-risk patients should be carefully counselled and offered supportive mental health resources throughout their cancer journey. Caring for a patient’s psychological well-being improves their quality of life, makes them more likely to adhere to medical recommendations and can also reduce the burden on the health care system by decreasing health care utilization.35,36
Future work may explore the relation between nonfatal self-injury and cancer stage. Rates of self-injury are likely to vary owing to different symptom burdens and prognoses associated with different cancer types and cancer stages.10 Our ability to analyze rates of nonfatal self-injury by cancer stage is currently limited, as stage is often missing from the OCR for systemic reasons.
The primary strength of this study lies in the cohort and study design, which strengthens the ability for causal inference. We adjusted for potential confounders, resulting in 2 comparable groups and accounted for pre-existing differences among the population. The longitudinal data, unique to our data sets, allowed us to capture rates of nonfatal self-injury for all individuals diagnosed with cancer in Ontario over a period of 12 years and allowed us to follow these individuals for up to 13 years after diagnosis. As our study takes place within a publicly funded health care system, loss of information owing to insurance status and loss to follow-up are minimal.
Limitations
One limitation of the study is that we likely underestimated the true incidence of self-injury by counting only nonfatal self-injury events that resulted in emergency department visits. However, collecting self-injury events from emergency department data has been shown to be an effective method to capture self-injury incidence23 and, as we use the same methods of data collection for both the cancer and control groups, our comparisons remain valid.
Conclusion
Individuals diagnosed with cancer are at increased risk for nonfatal self-injury compared with those without cancer, at least during the first year after diagnosis. Nonfatal self-injury is an important outcome of cancer-related mental health that must be considered when devising supportive care programs for patients with cancer. The findings from this study reinforce the need to provide robust and accessible psychosocial oncology programs to support mental health along the cancer journey, particularly in the first year after diagnosis, and highlight nonfatal self-injury as an important target outcome for potentially severe manifestations of poor mental health.
Footnotes
Competing interests: Antoine Eskander reports receiving grants from Merck and personal fees from Bristol Myers Squibb outside the submitted work. Julie Hallet reports receiving speaking honoraria from Ipsen and Advanced Accelerator Applications outside the submitted work. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Christopher Noel, Alyson Mahar and Rinku Sutradhar conceived and designed the study. Lena Nguyen, Julie Hallet, Antoine Eskander, Wing Chan, Christopher Noel, Alyson Mahar and Rinku Sutradhar acquired, analyzed and interpreted the data. Lena Nguyen, Julie Hallet, Alyson Mahar and Rinku Sutradhar drafted the manuscript. Antoine Eskander, Christopher Noel, Alyson Mahar and Rinku Sutradhar critically revised the manuscript for important intellectual content. Lena Nguyen and Wing Chan performed the statistical analysis. Julie Hallet and Antoine Eskander obtained funding. Alyson Mahar and Rinku Sutradhar supervised the study. Alyson Mahar and Rinku Sutradhar share co–senior authorship. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding: This work was supported by an AFP (Alternative Funding Plan) Innovation Fund.
Data sharing: The data set from this study is held securely in coded form at ICES. Data-sharing agreements prohibit ICES from making the data set publicly available.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care. Parts of this material are based on data and information provided by Cancer Care Ontario (CCO) and the Canadian Institute for Health Information (CIHI). The opinions, results, views and conclusions reported in this paper are those of the authors and do not necessarily reflect those of CCO or CIHI.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/2/E291/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors