Abstract
Background: Although the current Canadian Task Force on Preventive Health Care guideline recommends that physicians should inform women aged 40–49 years of the potential benefits and harms of screening mammography to support individualized decisions, previous reports of variation in clinical practice at the physician level suggest a lack of guideline-concordant care. We explored determinants (barriers and facilitators) of guideline-concordant care by family physicians regarding screening mammography in this age group.
Methods: We conducted qualitative semi-structured interviews by phone with family physicians in the Greater Toronto Area from January to November 2020. We structured interviews using the Theoretical Domains Framework to explore determinants (barriers and facilitators) of 5 physician screening behaviours, namely risk assessment, discussion regarding benefits and harms, decision or referral for mammography, referral for genetic counselling and referral to high-risk screening programs. Two independent researchers iteratively analyzed interview transcripts and deductively coded for each behaviour by domain to identify key behavioural determinants until saturation was reached.
Results: We interviewed 18 physicians (mean age 48 yr, 72% self-identified as women). Risk assessment was influenced by physicians’ knowledge of risk factors, skills to synthesize risk and beliefs about utility. Physicians had beliefs in their capabilities to have informed patient-centred discussions, but insufficient knowledge regarding the harms of screening. The decision or referral for mammography was affected by emotions related to past patient outcomes, social influences of patients and radiology departments, and knowledge and beliefs about consequences (benefits and harms of screening). Referrals for genetic counselling and to high-risk screening programs were facilitated by their availability and by the knowledge and skills to complete forms. Lack of knowledge regarding which patients qualify and beliefs about consequences were barriers to referral.
Interpretation: Insufficient knowledge and skills for performance of risk assessment, combined with a tendency to overestimate benefits of screening relative to harms affected provision of guideline-concordant care. These may be effective targets for future interventions to improve guideline-concordant care.
The lifetime risk of breast cancer among women in Canada is 1 in 9, with about 20% of cases occurring in women younger than 50 years.1 For women aged 50–74 years who are of average risk for breast cancer, the Canadian Task Force for Preventive Health Care (CTF-PHC) and the United States Preventive Services Task Force (USPSTF) recommend screening mammography every 2–3 years based on evidence of reduced risk of death from breast cancer.2–4 Both organizations recommend against routine screening for women aged 40–49 years owing to concerns that the harms (e.g., psychological effects, false-positive results, overdiagnosis) outweigh the benefits; however, they state that the decision to screen in this age group should be individualized, based on benefits, harms and patient values.2,4 Radiological societies advocate that routine screening should start at age 40 or 45 years.5–7
Providers report variation in practice patterns for women aged 40–49 years, with some always ordering screening mammography and others reporting that screening in this age group is unnecessary;8–10 this variation has been objectively confirmed at the provincial level in Ontario.11 Further, both the CTFPHC and USPSTF guidelines are for women of average lifetime risk for breast cancer, requiring physicians to discern which women are at higher than average risk for breast cancer and provide these patients with separate screening recommendations. This requires that physicians take a complete family history, evaluate for genetic (i.e., inheritable) risk and encourage high-risk screening, if required. In Ontario, eligibility criteria exist for free access to genetic testing if the chance of harbouring a BRCA mutation is greater than 10%.12
Providers report lack of support, time, absence of reminder services and confusion regarding conflicting evidence as barriers to screening for breast cancer.9,13 Many providers report never having referred a woman for genetic counselling or to a high-risk screening program.14,15
Variation in practice by providers would suggest provision of care that is not concordant with current guidelines, given that the behaviour should be based on a patient’s risk profile and values. Little is known about the underlying barriers and facilitators that could determine provision of guidelineconcordant care for women aged 40–49 years. Understanding these determinants is an important first step to selecting appropriate implementation strategies, as described in the knowledge-to-action framework.16 We explored determinants (including barriers and facilitators) of guideline-concordant care among family physicians, with a focus on 5 important screening-related behaviours necessary for guideline concordance in this age group.
Methods
Study design
We conducted 1-on-1 semi-structured interviews with a sample of primary care physicians in the Greater Toronto Area, Canada, between January and November 2020. We used a pragmatic, deductive qualitative approach, following guidance for using the Theoretical Domains Framework (TDF), a commonly used, theory-informed approach to comprehensively consider barriers and facilitators in implementation science.17,18 We followed the Consolidated Criteria for Reporting Qualitative Research (COREQ) checklist.19
Setting
The Ontario Health Insurance Plan (OHIP) is a publicly funded health insurance program for medically necessary care. For women in Ontario aged 40–49 years, a screening mammogram is covered by OHIP if it is accompanied by a physician referral, generally from their primary care provider or family physician.20 Therefore, family physicians are the gate-keepers of access to screening mammography in this age group. If a patient meets certain criteria (e.g., family history, ethnic background) that classifies them at higher than average risk, the physician should offer a referral to a genetic counsellor to assess eligibility for genetic testing or, for patients with a lifetime risk of breast cancer greater than 25%, a referral to the Ontario high-risk breast screening program.21 Patients in the high-risk screening program are offered OHIP-insured annual screening mammography and breast magnetic resonance imaging.
Study framework
The study team created an operational definition of guideline-concordant mammography screening in this age group based on the Canadian Task Force guideline,2 the associated Task Force tools22 and the Action, Actor, Context, Target, Time framework for specifying behaviours for the purposes of implementation work.23 We considered 3 behaviours necessary to fulfill guideline-concordant care, namely breast cancer risk assessment; discussion of benefits, harms and preferences (i.e., shared decision-making); and decision or referral for screening mammography. If the physician determined that screening was appropriate (benefits likely greater than harms) but the patient chose not to screen, this was considered concordant with guidelines, as patients have the right to decline investigations. If screening was inappropriate (harms likely greater than benefits), we considered the concordant behaviour as the physician not encouraging screening; however, if the patient expressed a desire for screening, a referral may still be considered guideline concordant so long as the physician elicited patient values, provided education and obtained comprehensive informed consent. Although not specifically part of the guidelines, we considered 2 additional behaviours as part of standard care given a positive family history, namely referral to genetics counselling and enrolment in high-risk screening.
Participants and recruitment
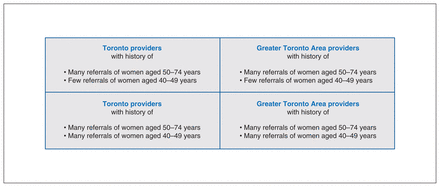
We used purposeful sampling, stratified by referral pattern and geographic location, to ensure inclusion of a diverse range of perspectives (Figure 1).24,25 A list of physicians who referred a patient for mammography in 2018 was generated by the Joint Department of Medical Imaging (JDMI). The JDMI is affiliated with most academic institutions in downtown Toronto but also accepts referrals from a wide catchment area outside Toronto. We scanned the list to identify family physicians practising in different geographic locations and rural areas to try to increase representation from these areas. We organized physicians by high and low referral rate by comparing their number of referrals for women aged 40–49 years and 50–74 years. We used these referral patterns only to ensure recruitment of diverse participants. Sampling was primarily within the Greater Toronto Area as we assumed that the barriers and facilitators there (which includes both high- and low-resourced areas) would not be substantially different from other high- and low-resourced cities in Ontario.
Categories for stratified purposeful sampling. “Referrals” describe the general pattern of family physician referrals to the Joint Department of Medical Imaging in Toronto, Canada. It was used for recruitment purposes only and does not objectively capture total physician referrals. “Toronto” includes the urban areas of Toronto, Thornhill and North York; “Greater Toronto Area” includes the suburban areas, namely Orangeville, Vaughan, Scarborough, Brampton, Pickering and Ajax.
We scrambled the list to facilitate arbitrary recruitment and invited potential participants to join the study in batches of 100 (25 per recruitment category). We prepared and mailed letters, and sent nonresponders up to 2 reminders, each 3–4 weeks apart, following the Dillman method.26 Physicians were eligible if they held a licence to practise in Ontario and consented to participate.
Data sources
All project collaborators met to discuss the interview guide, which was then pilot tested by the lead author (M.B.N.) and 2 other members of the study team (L.D. and N.I.). We structured the interview guide around the 5 provider behaviours of interest, namely breast cancer risk assessment; discussion of benefits, harms and preferences (i.e., shared decision-making); decision or referral for screening mammogram; referral for genetic counselling; and enrolment in a high-risk screening program. For each behaviour, we designed questions to understand current practice and explore determinants (i.e., barriers and facilitators) of behaviour by domains of the TDF.27,28 For example, to assess the TDF domain “beliefs about consequences,” we asked, “What ... harms do you think about related to sending a woman in this age group for a screening mammogram?” After about 2–3 interviews, it became apparent that asking about the guidelines caused confusion; therefore, we modified the guide to ask about routine practice, and followed these with the TDF-based questions. The original semi-structured interview guide and the TDF domain guide are presented in Appendix 1 and Appendix 2, respectively, available at www.cmajopen.ca/content/10/4/E900/suppl/DC1.
Data collection
We asked physicians to complete a short demographic questionnaire online before scheduling the interview. We conducted interviews over the phone. These were recorded, transcribed by a third party and anonymized. The interviewer (M.B.N.) is a breast medical oncologist with moderate interview experience. She was motivated to understand the problem of provider variability in guideline concordance after seeing women experiencing harms, including women who should have been offered earlier or additional screening given their high lifetime risk of breast cancer, as well as women who were overdiagnosed because of unnecessary screening despite low lifetime risk of breast cancer. The interviewer conducted interviews with a neutral, open-ended, nonjudgmental tone, without any intonation about what was correct. Interviewers and interviewees had no previous relationships, knowledge about practice or other goals, and no interviewer characteristics were disclosed.
Given the multiple behaviours of interest, we estimated 10–12 interviews as the lower limit for saturation. We increased this lower limit to address dialogue quality, to improve the experience of the interviewer and to reach less well-represented physician groups (e.g., men, physicians practising in locations outside of central Toronto). We did not perform any repeat interviews. We determined that we had reached saturation by considering the concept of information power.29 We also relied on guidance for achieving data saturation for theory-based interview studies, which suggests conducting at least 6–10 interviews, with a stopping criterion of 3 interviews with no new information.30 Recruitment, data collection, transcription and analysis continued until we reached saturation in all relevant TDF domains.
Data analysis
Two members of the research team (M.B.N. and A.M.C.) independently coded interview transcripts using directed content analysis, applying individual TDF domains as deductive codes as previously described.17,18 These 2 team members coded transcripts first by behaviour of interest, and then by the identification and application of the relevant TDF code, coding to multiple domains where appropriate. The 2 coders compared the coded text of each transcript and discussed discrepancies between themselves or with the research team until reaching a consensus. We did not conduct participant checking, given the variability in the responses. We entered transcripts with finalized codes into NVivo software and used the matrix tool to generate sequences of quotes that applied to each behaviour of interest and each TDF code. First, we reviewed the less commonly applied TDF codes to assess for important (but infrequently discussed) determinants. Next, we reviewed more commonly used TDF codes to generate descriptive narratives for each behaviour, which outlined the determinants that directly influenced the key provider behaviours. We discussed all relevant codes and used these to create tables showing the direct barriers and facilitators for each behaviour. We did not return transcripts to participants for comments or corrections as data were clarified during the interviews.
Ethics approval
This study was approved by the Research Ethics Board at Women’s College Hospital (no. 2019-0141-E).
Results
Twenty providers expressed interest in participation. Of these, 2 were not interviewed as their demographic category was already saturated. Among the 18 providers interviewed, the mean age was 48 years and 13 (72%) identified as women. Five providers (28%) had high referral rates for women aged 40–49 years, and 9 (50%) had low referral rates for this age group, despite high rates for those 50 years and older (Table 1). Interviews lasted about 30–45 minutes.
Participant demographics
Typical practice and behavioural entry points
Primary care physicians described 2 situations that could start the 5 behaviours. The first was at a scheduled periodic health visit (i.e., complete physical or annual health exam). The appointment served as reinforcement for the risk assessment or other behaviours of interest, as this provided the occasion for providers to update the full family history. Thereafter, some physicians engaged in the remaining guidelineconcordant behaviours; however, others proceeded to discussion or referral only if the results of the risk assessment (complete or incomplete) were judged as higher than average. A subset of providers with strong intentions to screen — owing to the belief that screening mammography should be initiated at age 40 years for all women — described directly referring for screening mammography without risk assessment.
The second entry point to screening behaviours was the social influence of a patient asking about screening. This either started the behavioural sequence or led directly to physicians providing an explanation to patients that guidelines do not recommend screening until age 50 years. In these situations, it appeared that neither screening nor referrals to genetics counsellors or high-risk screening programs were considered because physicians did not review the family history, or discuss or assess risk.
Behaviour 1: risk assessment
Barriers
Barriers to an individualized risk assessment for breast cancer included knowledge of risk factors and of risk assessment tools, skills to synthesize risk factors or use the tools, and beliefs about consequences that the tools do not guide further management (Table 2). Physicians had difficulty listing risk factors for breast cancer aside from family history, such as reproductive factors, ethnicity or breast density. Some physicians lacked skills to calculate an overall risk of breast cancer. Physicians were confused about the difference between an individualized risk assessment that would prompt a discussion regarding mammography versus the family history criteria that should prompt a genetics referral. Some suggested that if women did not meet the criteria for high-risk screening (≥ 25% lifetime risk), that this was synonymous with not qualifying for early screening mammography.
Barriers and facilitators of risk assessment
Beliefs about consequences were related to the physicians’ environment, context and available resources (such as a risk assessment tool). Physicians were often not aware of existing risk calculators, did not know how to use them or found them time consuming and impractical. They expressed concerns regarding their limitations, such as the fact that some risk factors (breast density) were not included. Physicians pointed out that the risk calculation was not tied to any management recommendation. They also described how no intermediate risk management option was available, as with other primary care stratification tools, such as the Framingham for cardiovascular disease31 or FRAX fracture risk assessment tool;32 these tools provide 3 risk strata with recommended management strategies associated with each strata.
Facilitators
Physicians stated that more explicit recommendations within the guidelines regarding the need for risk assessment and the recommended tool would be helpful. They noted that a simple, user-friendly tool that listed all important risk factors would be within their professional role and scope to complete, and that they had the skills to use an online tool or application. Some stated that, if the tool had a checklist of risk factors and could be embedded into their electronic medical record, this could help with knowledge and reinforcement of relevant risk factors.
Behaviour 2: discussion about mammography benefits and harms
Facilitators
The discussion about benefits and risks of screening was facilitated by physicians feeling it was their professional role to provide patients with as much accurate information as possible to inform their decision. Many stated that they were accustomed to having discussions with patients about benefits and harms of a test or procedure, as these types of discussions are prevalent for other screening tests in primary care. Some physicians expressed that they were confident in their ability to explain to specific patients why screening was not recommended routinely. Physicians advocated for the maintenance of the periodic health visit to facilitate review of family history and assessment or discussion regarding screening for the major cancer types (Table 3).
Barriers and facilitators of discussion
Barriers
Some physicians appeared not to discuss all of the pertinent harms of screening mammography. This occurred either because of a lack of knowledge of mammography harms or their belief that the information about potential harms would lead a woman to decide against screening. Knowledge among physicians was specifically low regarding the harm of overdiagnosis. Many believed that screening mammography was similar to a call-back screen, biopsy or pre-cancer (ductal carcinoma in situ) diagnosis, which limited a comprehensive, informed discussion.
Behaviour 3: Decision or referral for guideline-concordant screening decisions
We observed 3 common physician patterns when exploring decisions or referrals for screening mammography, in accordance with guidelines. Physicians with strong intentions to screen sent screening referrals in response to emotion (e.g., with regard to outcomes of past patients), the social influence of patients (e.g., their concerns about risk of cancer) or the social influence of radiologists. In contrast, physicians who interpreted that the guidelines stated not to screen until age 50 years had strong beliefs in their capabilities to educate patients about why screening was not recommended and did so. A third group of physicians performed the risk assessment and discussion, and based their final recommendation regarding whether or not to screen on their beliefs about consequences; however, they tended to overestimate the benefits or underestimate the harms of screening. This knowledge gap contributed to a (potentially unwarranted) perception that their own screening referrals were concordant with guidelines (Table 4).
Barriers and facilitators of decision or referral for mammography
Facilitators
Some physicians had knowledge, skills and beliefs in their capabilities to explain to patients why screening was not routinely recommended. In addition, some radiology departments accepted referrals only if primary care providers clearly documented patients’ increased risk of breast cancer, which reinforced guideline-concordant referrals.
Barriers
Physicians with strong intentions to screen were primarily influenced by emotion. They described previous experience of patients in this age group with clinically detected, rather than screen-detected, cancer and drew the (potentially inappropriate) conclusion that the outcome would have been different had these patients engaged in screening. Others sought to avoid regret related to recommending against screening for patients who may eventually develop breast cancer. Providers also cited the social influence of radiology guidelines or radiologists, describing that they would have the most accurate information. The environment, context and resources of radiology departments who routinely accepted these referrals reinforced guideline-disconcordant decisions. Some wondered if the guidelines were based on cost considerations, rather than optimal patient care.
Physicians who tended to overestimate the benefits and underestimate the harms of screening also tended to refer for screening. Knowledge gaps included the assumption that it was always better to “catch something earlier” and an incomplete understanding of screening harms. Many discussed risks of discomfort and radiation, but did not comment on frequency of false-positive results or the concerning clinical impact of overdiagnosis. We also observed additional beliefs about consequences regarding the financial impacts or time burden of screening on the patient. Physicians noted that marginalized populations, such as those living in remote communities or those without the ability to take paid time off work, were more at risk of not attending their appointments.
Behaviours 4 and 5: Referral to genetic counselling and enrolment in high-risk screening program
Physicians described similar practice patterns regarding the behaviours of genetics referral and enrolment of patients in the Ontario high-risk screening program. After elicitation of family history, some physicians referred patients with notable family histories to “high-risk breast clinics” or “genetics centres,” which provided comprehensive assessment and managed several aspects of care, including referral for genetic counselling, enrolment in the high-risk screening program and sending recommendations back to the primary care provider regarding early screening mammography (Table 5).
Barriers and facilitators of referral to genetic counselling and to high-risk screening programs
Facilitators
Providers who referred to these centres described them as an excellent environmental resource, one that they could rely on to manage comprehensive patient care and provide advice to providers about ongoing management. On review of physician practice locations, those who had access to these centres appeared to be in areas of higher socioeconomic status, with access to academic centres (i.e., high-resource areas). For physicians aware of Cancer Care Ontario’s referral forms, the environment acted as a facilitator; using the listed criteria on the forms, providers described a belief in their capability to identify the correct patients and complete the forms.
Barriers
Providers who appeared unaware of or who did not have access to these comprehensive centres described barriers related to environment and beliefs about genetic clinics not accepting referrals from community physicians, the cumbersome nature of paperwork and forms, and that patients would fall through the cracks. These barriers were exacerbated by patient factors, such as the patient not knowing their complete family history, difficulty finding transportation or financial constraints to attend the appointment. Although physicians felt it was their role to identify patients with notable family histories and provide a referral, they described the gap in their knowledge and skills, such as being unaware of the criteria for genetic testing. These providers stated a checklist would help facilitate referrals, but appeared unaware that a checklist existed on a standard referral form listed on the provincial website.
Interpretation
In this study, we unpacked the determinants for family physicians’ approaches to 5 behaviours necessary for guidelineconcordant screening for breast cancer in women aged 40–49 years. The behavioural sequence was often triggered by a periodic health visit or a patient-initiated conversation. Barriers to risk assessment included knowledge of risk factors and risk assessment tools, skills to synthesize risk and beliefs that the tools do not help guide management. Providers felt confident with the general practice of shared decision-making; however, they had insufficient knowledge and sometimes incorrect beliefs about consequences, which limited a fully informed discussion. Emotion, the social influence of patients and radiologists, knowledge and beliefs about the benefits and harms of screening influenced guideline-disconcordant referrals for screening. Referrals to genetic counselling or high-risk screening programs were facilitated by the environment, namely access to centralized clinics; however, barriers included lack of knowledge and skills about referral criteria.
The benefits of screening are determined by an individualized risk assessment. Previous qualitative studies have reported similar findings of provider-level barriers related to knowledge of risk factors, skills to consider multiple risk factors and perceptions of cumbersome risk assessment tools.33,34 An additional requirement for risk assessment is the ability to gather an accurate and comprehensive family history. Although physicians in our study described thoroughly and routinely collecting family history, the literature suggests that this may occur less consistently than assumed.35 Further, physicians described that limited information within the guidelines could be acted on for risk stratification, in keeping with previous reports that highlighted a lack of decision-support tools to help physicians make shared decisions with patients.36
Variation in practice has been attributed to differences in beliefs regarding the efficacy of mammography.10,37 Our data expand upon this, suggesting providers may incorrectly evaluate or misrepresent the balance between benefit and harm either by overestimating the benefit, underestimating the harms or both. Research on cancer screening in general suggests that primary care providers are more likely to order screening tests when patients display anxiety about cancer, patients have expectations about receiving tests or providers believe screening has more benefits than harms.38
Breast cancer that is detected clinically (without screening) in a woman in her 40s could be misinterpreted as a situation in which the outcome would have been different if she had engaged in routine screening, although this is not necessarily the case. This can lead to overestimation of the benefits of screening, feelings of regret and increased recommendations for screening.39 This cognitive bias is termed “loss aversion bias,” which describes an individual’s tendency to prefer avoiding losses than acquiring equivalent gains. It can result in misestimation of benefit (i.e., avoiding “missing” a cancer) and has been shown elsewhere in medicine.40 In behavioural science, this anticipated regret is strongly correlated with intentions and behaviour.41
Physician underestimation of screening harms was related to insufficient knowledge; physicians did not communicate harms because of the belief that it would lead patients to decide against screening. Previous studies have found that when women are told about the harms of screening, particularly the possibility of overdiagnosis, this can change their attitudes and intentions to screen.42 Lack of accurate communication regarding screening harms is prevalent in patient education materials,43–45 showing another way by which the environment influences patient and provider knowledge. This omission speaks broadly to the ethical implications of an informed decision.46 The medical ethical principle of respect for patient autonomy affirms the right of patients to the information necessary to make decisions and, therefore, the obligation of health professionals to provide this information to patients.47 Overcoming the barriers to underestimation of harms will increase the likelihood of a more accurate estimation of the benefit-to-harm ratio to guide discussions and decisions, which could improve the variation in practice.
Family physicians described that some radiology departments accepted all, none or only select referrals for screening mammography and that this reinforced their behaviour, appropriately or not. The differences in radiology departments is corroborated by a recent study that showed that up to 80% of radiology department decisions differed from the USPSTF recommendations,48 creating confusion among providers about optimal referral behaviour. This suggests that an intervention to standardize practice or referral forms for physicians to communicate to radiologists that risk assessment and informed shared decision-making discussion have been performed could help to reinforce guideline-concordant behaviour.
The TDF framework allows mapping of these behavioral determinants to behaviour change techniques to inform interventions that may increase guideline-concordant behaviour.49,50 Important behaviour change techniques may include information regarding the behaviour, persuasive communication, rehearsal of relevant skills and training. Important skills and training include use of a risk assessment calculator. To target emotion and social influences, behaviour change techniques of coping, planning, cognitive restructuring and modelling would likely be helpful, with a focus on addressing the misunderstanding that all breast cancers detected clinically could have been prevented by screening. These techniques should also target provider knowledge and ability to communicate the concept of length time bias to patients (i.e., overestimation of survival duration).
Limitations
Guideline-concordance could not be confirmed, but only inferred through listening and analysis of the physician’s approach. We limited our recruitment to 1 major urban centre, predominantly interviewed female family physicians and did not interview rural physicians, potentially contributing to participation bias and, therefore, generalizability of results. Despite these limitations, physicians described variation in practice and were forthcoming with answers with regard to approaches, barriers and knowledge gaps. It did not appear that the barriers (with the exception of environment) were specific to practice location, nor that they would be different in other major urban Ontario cities. The literature suggests that female physicians are more likely to order more screening tests (mammography, pap smears),51 but further research and analysis is required to understand if women experience different barriers or facilitators than men. The axiology of the primary researcher should be considered; M.B.N. prioritized understanding the problem over any personal opinions about screening. Other forms of qualitative inquiry, such as direct observation or document analysis, may have revealed other barriers or facilitators. The confirmatory data in the literature suggest that our results are credible and confirmable, and that our key findings are transferrable to others working in similar health care systems in which women are insured for screening services.
Conclusion
Guidelines state that physicians should make individualized decisions regarding screening mammography with women aged 40–49; however, barriers of knowledge, skills, beliefs about consequences, environment, emotion and social influences affected the provision of guideline-concordant care. Overall, interventions to target knowledge and skills related to risk assessment, knowledge and awareness of benefits and harms; improved guideline clarity with decision-making support; and policy changes regarding radiology departments may improve guideline concordance.
Footnotes
Competing interests: Alexandra Desnoyers reports consulting fees from Novartis and Pfizer, and payments from Novartis, Pfizer, AstraZeneca, Gilead and Apobiologix. Eitan Amir reports consulting fees from Sandoz, Novartis and Exact Sciences. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Michelle Nadler, Laura Desveaux, Eitan Amir and Noah Ivers conceived and designed the study. Michelle Nadler collected data. Michelle Nadler, Ann Marie Corrado, Laura Desveaux, Sarah Neil-Sztramko, Eitan Amir and Noah Ivers contributed to data analysis. All of the authors contributed to interpretation of data. All of the authors drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: Michelle Nadler was supported as a Dream Hold ‘Em For Life clinical oncology fellow. Ann Marie Corrado was supported by the Peter Gilgan Centre for Women’s Cancers at Women’s College Hospital, in partnership with the Canadian Cancer Society. Brooke Wilson was supported as a National Breast Cancer Foundation of Australia International Fellow. Alexandra Desnoyers was supported as a Dream Hold ‘Em For Life clinical oncology fellow. Noah Ivers is supported by a Tier 2 Canada Research Chair in implementation of evidence-based practice and as a clinician scientist by the Department of Family and Community Medicine, University of Toronto.
Data sharing: Data are not available for use by other authors.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/4/E900/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors