Abstract
Background: The debate over acetylsalicylic acid (ASA) therapy for primary prevention of cardiovascular disease (CVD) has recently resurfaced, but scarce data are available on prophylactic ASA use in Canada for this purpose. This study aimed to evaluate the prevalence and factors associated with ASA use, and the potential impact of implementing the most recent (2016) US Preventive Services Task Force recommendations for primary CVD prevention in a Canadian setting.
Methods: We performed a cross-sectional analysis using data from the CARTaGENE study, which included a representative sample (n = 20 004) of the 2018 general population of the province of Quebec. We assessed eligibility for ASA treatment using US Preventive Services Task Force criteria (age 50–69 yr, no past history of myocardial infarction or stroke, and 10-year risk of CVD of at least 10%). We extrapolated to the entire 2018 Quebec population the number of people who would need to start ASA treatment.
Results: A total of 6231 respondents in the CARTaGENE study (54.2% of those aged 50–69 yr with no prior history of CVD) were found to be potentially eligible for ASA use for primary CVD prevention. Of the 6231, 1379 (22.1%) were receiving prophylactic ASA treatment. Factors found to be related to ASA use included age, male sex, regular medical visits, lower education level, obesity, hypertension, diabetes and dyslipidemia. Income and smoking status were not found to be significantly associated with ASA use. Our results indicate that 885 261 people would potentially have started ASA treatment if the US Preventive Services Task Force recommendations had been implemented in Quebec in 2018.
Interpretation: Prevalent ASA use for primary CVD prevention was low. Implementation of the 2016 US Preventive Services Task Force recommendations would require initiating ASA treatment in a substantial proportion of people, with undetermined potential benefits.
Cardiovascular disease (CVD) is the leading cause of mortality worldwide,1 accounting for 31% of all deaths in 2016. In Canada, CVD was responsible for 66 922 deaths in 2017.2 Improving cardiovascular health is a major target for research and prevention programs, and anti-platelet therapy has become one of the main strategies to reduce the risk of CVD. The benefits of low-dose acetylsalicylic acid (ASA) in secondary CVD prevention are well established.3,4 In primary prevention, however, there is no clear consensus on ASA use, as the evidence regarding its benefits remains uncertain. The debate over ASA therapy for primary CVD prevention has recently resurfaced, fuelled by trials such as ASCEND (A Study of Cardiovascular Events iN Diabetes)5 and ARRIVE (A Study to Assess the Efficacy and Safety of Enteric-Coated Acetylsalicylic Acid in Patients at Moderate Risk of Cardiovascular Disease),6 which showed minimal cardiovascular benefits from its use.
The American College of Cardiology and the American Heart Association recently issued a joint guideline on primary prevention of CVD in which they state that low-dose ASA might be considered for adults aged 40–70 years at higher risk for CVD who do not appear to be at increased risk for bleeding.7 Risk factors for bleeding that need to be assessed for include a history of previous gastrointestinal bleeding or peptic ulcer disease, bleeding at other sites, thrombocytopenia, coagulopathy, chronic kidney disease and concurrent use of other medications that increase bleeding risk, such as non-steroidal anti-inflammatory drugs, steroids, direct oral anticoagulants and warfarin. Likewise, the most recent (2016) US Preventive Services Task Force recommendations suggest initiating ASA treatment in adults aged 50–59 years with a 10% or greater 10-year CVD risk given that the benefits probably outweigh the risks in these people.8 For adults aged 60–69 years with increased CVD risk, the decision should be individualized, but the benefits probably slightly outweigh the risks according to these recommendations.8
Investigators in the United States and other countries have studied the prevalence of ASA use for primary prevention,9–12 but similar data are scarce in Canada. In the present analysis, we first evaluated the frequency of ASA use for primary CVD prevention and factors associated with its use in a large population-based survey. We then sought to evaluate the potential impact of implementing the 2016 US Preventive Services Task Force recommendations by extrapolating to the entire general population the number of people who would need to start ASA treatment.
Methods
Design and study population
We performed a cross-sectional study using data from 20 004 randomly selected people aged 40–69 years from the Quebec general population included in the CARTaGENE cohort.13 Given the growing burden of chronic disorders, the CARTaGENE cohort was set up as a large biobank and databank so that determinants of diseases, including environmental, lifestyle and genetic factors, could be uncovered. This was a complex sample survey, and, overall, the final cohort was representative of the general population when compared with characteristics obtained from the 2006 Canadian census. Comparison of sociodemographic characteristics between the CARTaGENE cohort and the general population showed good concordance except that CARTaGENE participants were slightly more educated and ethnic minorities were slightly overrepresented.13
Data sources
Participants were recruited from August 2009 through October 2010 by means of a provincial government health insurance database. Participants completed an extensive interviewer-administered questionnaire inquiring about health, lifestyle and sociodemographic characteristics at one of the assessment sites. Anthropometric measurements, blood pressure measurements and blood sampling were performed at the time of the interview. Additional details on questionnaire domains, with sources of data, physical measurements and biochemical measures that were used for the study, are provided in Appendix 1 (available at www.cmajopen.ca/content/8/1/E41/suppl/DC1). A detailed description of CARTa-GENE methods has been published elsewhere.13
Eligibility for acetylsalicylic acid therapy
All participants were evaluated for eligibility for ASA therapy for primary CVD prevention. Cardiovascular disease was defined as a self-reported history of myocardial infarction, angina or stroke. For all participants without a history of CVD, 10-year CVD risk was estimated with the Framingham Risk Score,14 taking into account the participant’s age, sex, total and high-density lipoprotein cholesterol values, systolic blood pressure, use of antihypertensive drugs, presence of diabetes and smoking status. Based on the 2016 US Preventive Services Task Force recommendations,8 participants aged 50–69 years with no past history of myocardial infarction or stroke and with a 10-year CVD risk of at least 10% were considered eligible for daily ASA use. Participants for whom a Framingham Risk Score could not be calculated because of missing data were excluded from the analyses, but multiple imputation for missing values was performed to evaluate the impact on study results. We also analyzed the number of participants taking anticoagulant drugs to estimate the number of candidates eligible for ASA treatment who might have a contraindication to its use. We did not exclude these participants from the analyses. Finally, we estimated the number of people who would need to start ASA treatment by extrapolation to the Quebec population using 2018 population estimates.15
Measures
Information was collected for all participants on the presence of a self-reported history of CVD, hypertension, diabetes or dyslipidemia, and for corresponding treatments (i.e., use of antihypertensive drug[s], antidiabetic agent[s] or statin[s]). Participants were asked to give the name of all prescribed and nonprescribed drugs they were using at the time of the interview and to provide, if possible, the drug containers so the information could be validated. For analyses, participants who reported use of antiplatelet agents other than ASA were included in the ASA users’ group. Information on self-reported regular medical visits was also obtained; a regular medical visit was defined as at least 1 routine medical examination in the previous year.
Sociodemographic variables collected included age, sex, income and education. Annual income was stratified into 3 categories: low (< $50 000), medium ($50 000–$150 000) and high (> $150 000). Similarly, 3 education categories were created: low (high school or less), medium (college) and high (university).
Blood pressure values were obtained by calculating the mean of 3 measurements taken with an automated device by means of an oscillometric method. Blood samples were collected for lipid profile measurement, used to compute the Framingham Risk Score. Obesity was defined as a body mass index of 30 or greater. Smoking status was classified into 4 categories: never smoked (lifetime smoking history < 100 cigarettes), active smoker (≥ 1 cigarette daily in previous 30 d), occasional smoker (≥ 1 cigarette in previous 30 d) and past smoker (no cigarette in previous 30 d).
Statistical analysis
Primary analyses were restricted to the subgroup of participants eligible for antiplatelet treatment for primary CVD prevention, as defined in the US Preventive Services Task Force recommendations.8 We determined factors associated with ASA use using logistic regression. We performed χ2 tests for categorical variables and trend tests for ordinal variables. We conducted logistic regression analyses to identify variables related to ASA use. Variables that were included in the model were age, sex, regular medical visits, education, income, smoking status, obesity, hypertension, diabetes and dyslipidemia. We assessed discrimination of the model by the C-statistic and calibration by the Hosmer–Lemeshow goodness-of-fit test. Statistical tests were 2-sided, and a p value < 0.05 was considered to represent statistical significance. In the subgroup of participants with no history of CVD, we performed multiple imputation (chained equation imputation) for missing values of the following variables: Framingham Risk Score, education, smoking, obesity and annual income. We performed all analyses using SPSS software, version 24 (IBM Corporation).
Ethics approval
The study was approved by the Hôpital du Sacré-Coeur de Montréal Research Ethics Committee. All CARTaGENE participants signed a consent form, and the study adhered to the Declaration of Helsinki.
Results
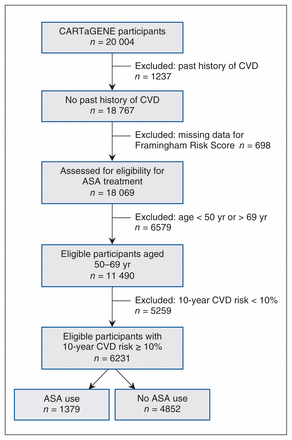
The eligibility of CARTaGENE participants for ASA use for primary CVD prevention is shown in Figure 1. Of the 20 004 participants, we excluded 1237 (6.2%) with a past history of CVD, of whom 504 (40.7%) reported a history of myocardial infarction alone, 274 (22.2%) stroke alone, 55 (4.4%) both, and 404 (32.7%) angina alone. Regular use of ASA was reported by 906 (73.2%) of those with prior CVD. We excluded participants for whom the Framingham Risk Score could not be calculated owing to missing data (n = 698).
Applying the US Preventive Services Task Force eligibility criteria identified 6231 potential candidates for ASA use in primary CVD prevention (54.2% of the 11 490 participants aged 50–69 years with no past history of CVD) (Figure 1). The prevalence of antiplatelet therapy was low among eligible participants (1379 [22.1%]). Of the 1379, 21 (1.5%) reported using clopidogrel instead of ASA, and 9 reported using both ASA and clopidogrel. Of the 4852 participants not receiving any antiplatelet agent, 58 (1.2%) were receiving an anticoagulant medication, which contraindicates the use of ASA for primary CVD prevention.
Key baseline characteristics of CARTaGENE participants eligible for preventive ASA use according to US Preventive Services Task Force recommendations are presented in Table 1. Eligible candidates had a mean age of 60.1 (standard deviation 5.7) years, and the majority (4489 [72.0%]) were men. A total of 787 participants (12.6%) reported a diagnosis of diabetes, 2512 (40.3%) a diagnosis of hypertension, and 2375 (38.1%) a diagnosis of dyslipidemia.
Baseline characteristics of CARTaGENE study13 participants eligible for acetylsalicylic acid therapy for primary prevention of cardiovascular disease
Table 2 presents factors associated with ASA use. There were significant associations between ASA use and all studied variables except for sex. Of the 787 eligible participants with diabetes, 478 (60.7%) reported using ASA, and a majority of these participants were at high risk for CVD (Framingham Risk Score ≥ 20% in 325 [68.0%], obesity in 272 [56.9%], hypertension in 319 [66.7%] and dyslipidemia in 337 [70.5%]).
Acetylsalicylic acid use according to demographic and clinical characteristics
Multivariate analyses performed to identify factors associated with ASA use showed that ASA use was positively associated with older age, male sex, regular medical visits, lower education level, obesity, hypertension, diabetes and dyslipidemia (Table 3). Income and smoking status were not found to be significantly associated with ASA use. When these analyses were repeated after multiple imputation for missing values, there were no meaningful differences identified in the risk factors or the odd ratios.
Factors associated with acetylsalicylic acid use for primary prevention of cardiovascular disease among eligible participants on multivariate analysis
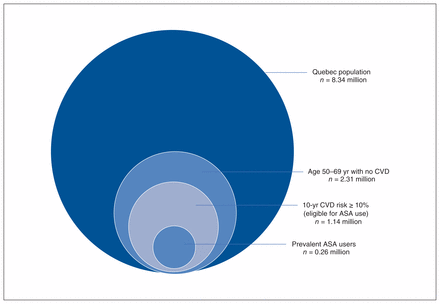
Our results indicate that 885 261 people would potentially have started ASA treatment if the US Preventive Services Task Force recommendations had been implemented in Quebec in 2018 (Figure 2).15
Estimate of the Quebec population eligible for acetylsalicylic acid (ASA) use for primary prevention of cardiovascular disease (CVD) according to the 2016 US Preventive Services Task Force recommendations8 and prevalent users. Implementation of the recommendations would necessitate initiation of ASA treatment in an additional 0.88 million people (1.14 million eligible people −0.26 million prevalent ASA users).
Interpretation
Using US Preventive Services Task Force criteria8 in a large representative population-based sample from Quebec, we found that a majority of people from the targeted age group with no prior CVD history were eligible for preventive ASA use but that, among these participants, the prevalence of ASA use was low (22%). Implementation of these recommendations in Quebec would thus require initiating ASA for a substantial number of people. We identified factors associated with ASA use, some of which (high blood pressure, diabetes and dyslipidemia) increased the odds of prophylactic ASA use more than twofold.
Other estimates of preventive ASA use in various cohorts have previously been published, with rates as low as 3% in a Swedish cohort of 5725 participants16 and as high as 47% in a US Web-based survey.12 In a study by Fiscella and colleagues9 using National Health and Nutrition Examination Survey data, 87% of men aged 45–79 years and 16% of women aged 55–79 years were eligible for antiplatelet therapy according to the 2009 US Preventive Services Task Force recommendations, 17 but only 32% of eligible men and 47% of eligible women reported current ASA use. The prevalence of ischemic vascular disease in the CARTaGENE sample (6.2%) is equivalent to that reported in a Canadian survey18 but somewhat lower than that reported in surveys from the US with similar designs.10–12
Multiple factors may explain the low rate of ASA use in the present survey. First, recommendations on ASA use for primary CVD prevention were conflicting at the time of the study (2010) and still are today (Table 4). For instance, in contrast to the 2016 US Preventive Services Task Force recommendations, 8 the US Food and Drug Administration does not support ASA use as a primary prevention strategy to reduce the burden of CVD.21 In its 2011 guideline, the Canadian Cardiovascular Society did not recommend the routine use of ASA but suggested that it could be considered for those at high vascular risk.25
Summary of recommendations for acetylsalicylic acid use in primary prevention of cardiovascular disease
What explains such discrepancies between guidelines? Large meta-analyses of primary prevention trials3,26–30 suggest that ASA lowers the risk of major CVD events (hazard ratio 0.89, 95% confidence interval 0.84–0.94),29 which is driven mainly by a reduction in the risk of nonfatal myocardial infarction. Studies have also shown potential long-term benefits of ASA use on colorectal cancer incidence and mortality when taken for at least 10 years.31 A recent meta-analysis, however, did not show any reduced incidence of colorectal cancer.29 The net impact of these results in terms of absolute benefits depends on the baseline risk of ischemic vascular events, and these potential benefits must be weighed against the inherent risks associated with daily ASA use, mainly risk of bleeding (hazard ratio 1.43, 95% confidence interval 1.30–1.56).29 More recent data from the ASCEND5 and ARRIVE6 trials are consistent with these previous trials. The relative benefits of daily ASA use compared to risks appear limited in recent trials,5,6 leading to a lower class of recommendation in the latest American College of Cardiology/American Heart Association guideline.7
Weighing benefits and harms of ASA use in primary prevention of CVD for a given patient undoubtedly remains a challenge. One major finding of recent trials is the overestimation of CVD risk from risk calculators. For instance, in the ARRIVE trial,6 which aimed to study prophylactic ASA use in a population at moderate risk for CVD, total events were lower than predicted by risk scores. Further studies will need to strive to identify more accurate ways of defining a subpopulation of patients with increased baseline CVD risk who would likely benefit from ASA use.
Limitations
There are some limitations to our study. The methodology for recruitment of CARTaGENE participants may have introduced a selection bias; however, overall, the final cohort was shown to be representative of the general population of the province of Quebec. Moreover, many variables were self-reported, as is the case in most similar studies, which potentially limits the exactitude of collected data. For instance, the presence of CVD was self-reported, which may have led to some misclassification. However, unlike other researchers, the CARTaGENE investigators included extensive data from biologic sampling and physiologic measurements, so that CVD risk scores could be calculated for most respondents.
The detailed questionnaire completed by participants could not capture cases in which a deliberate decision was made by a physician not to prescribe ASA after evaluation of benefits and harms of such a treatment. In some instances, this decision might also have originated from the participant’s preferences. Similarly, for respondents who reported regular antiplatelet use, the questionnaire could not ascertain the main purpose of such use, which might not always have been primary CVD prevention. Furthermore, our evaluation of potential contraindications to antiplatelet therapy was incomplete and our assessment of bleeding risk limited. However, we explored data on anticoagulation, which showed that only 1.2% of people at high risk not receiving preventive ASA were taking an anticoagulant agent and thus had a contraindication to ASA use for primary CVD prevention.
When enrolment in CARTaGENE began, different guidelines were in place. For instance, the 2009 US Preventive Services Task Force recommendations17 differed from the updated 2016 recommendations8 in that preventive ASA use was recommended based on different proposed cut-offs of 10-year CVD risk estimates based on age and sex. Finally, there were missing data. However, the proportion of missing values for most variables was very small, and when the analyses were repeated after multiple imputation for missing values, there were no meaningful differences identified in the risk factors or the odd ratios.
Conclusion
We showed that the use of ASA for primary CVD prevention was low in a large Canadian population-based survey. Strict implementation of 2016 US Preventive Services Task Force recommendations would require initiating ASA treatment in a substantial proportion of people, with undetermined potential benefits.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: François Madore supervised the project. Myriam Khalili, Fanny Lepeytre and François Madore conceived the study. Myriam Khalili, Fanny Lepeytre, Jason Robert Guertin and François Madore designed the study and analyzed the data. Myriam Khalili, François Madore and Jason Robert Guertin drafted the manuscript. All of the authors contributed to interpretation of data, revised the manuscript critically for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This work was supported by personal research funds from the authors. CARTaGENE was supported by Genome Canada, Génome Québec and the Quebec Ministry of Health. Rémi Goupil and Jason Robert Guertin hold research scholarships from the Fonds de recherche du Québec – Santé.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/8/1/E41/suppl/DC1.
References
- Copyright 2020, Joule Inc. or its licensors