Abstract
Background: International studies have observed inequities in stage at diagnosis of melanoma. As this has not been sufficiently studied in Canada, the purpose of this study was to investigate whether there are disparities in the diagnosis of advanced-thickness melanoma in the province of Ontario.
Methods: In this retrospective population-based cohort study, we obtained, abstracted and linked pathology reports for a 65% random sample of all cases of invasive cutaneous melanoma in Ontario from 2007 to 2012 in the Ontario Cancer Registry. Cases without pathology reports or with unreported thickness were excluded from the primary analysis. Associations between advanced melanoma (thickness > 2.0 mm) and patient, health-system and tumour factors were described and analyzed using multivariable modified Poisson regression.
Results: In total, 8042 patients had histologically confirmed melanoma and thickness information. Of these, 46.7% (n = 3755) were female, the median age at diagnosis was 62 years and 25.7% (n = 2069) had advanced melanoma. In multivariate analyses, advanced age (relative risk [RR] 1.53; 95% confidence interval [CI] 1.37–1.72), male sex (RR 1.12, 95% CI 1.05–1.20), lowest socioeconomic status quintile (RR 1.24; 95% CI 1.12–1.38) and health region (RR range 0.92–1.34, p = 0.005 for variable) were significantly associated with advanced melanoma. Presence of ulceration significantly modified many of these associations.
Interpretation: Disparate rates of advanced melanoma according to patient and health system factors suggest there may be inequitable access to timely diagnosis of melanoma in Ontario. This highlights a potential opportunity for system improvement to ensure timely and equitable access to melanoma care.
Canadian studies investigating disparities in melanoma thickness at diagnosis have rarely been performed because of the challenges in collecting melanoma stage information at the population level. Studies conducted in other jurisdictions have found disparate rates of advanced melanoma according to race/ethnicity,1–3 socioeconomic status (SES),4–6 age,7–10 sex,7,10 anatomic site,11,12 histological subtype13,14 and area of residence.15 As many studies were conducted in a setting without publicly funded universal health care (i.e., the United States), their results may not be generalizable to populations where universal health care exists.
We set out to evaluate patient and health-system factors that are independently associated with advanced melanoma diagnosed in the Canadian province of Ontario. We will also describe the impact of ulceration on identified relationships, hypothesizing that this feature would influence the ease of early detection of melanoma in our cohort.
Methods
Study design
This was a retrospective population-based cohort study. The cohort was selected to provide a representative picture of disparities in the diagnosis of advanced cutaneous melanoma across Ontario.
Study population
The study was conducted using a 65% random sample of all invasive melanoma cases diagnosed in Ontario between Jan. 1, 2007, and Dec. 31, 2012, in the Ontario Cancer Registry (OCR). The OCR is administered by Cancer Care Ontario, the provincial cancer agency associated with Ontario’s single-payer universal health system. The random sample was a convenience sample based on power requirements for the parent study investigating melanoma treatment outcomes according to stage. Patients whose first melanoma diagnosis was purely in situ on all specimens were excluded, because of the possibility of greater screening in these individuals and the fact that pure in situ data in the OCR are probably incomplete. Patients determined to be from out of province and those without a pathology report from Cancer Care Ontario were excluded. Details of the earliest melanoma were used when multiple primaries were reported. Patients younger than 20 years of age were also excluded.
Data sources
Ontario Cancer Registry and pathology reports
Data from Cancer Care Ontario’s population-based OCR were used to identify cases of melanoma. This registry is known for its very high level of accuracy and completeness levels of 95% overall.16 Data contained in the OCR include patient demographic characteristics and stage information on a subset of patients seen in cancer centres and at other selected health care sites.
Available pathology reports for all patients were provided by Cancer Care Ontario, abstracted according to a standardized algorithm and deterministically linked to each patient’s OCR record according to their group identification. Reliability testing indicated 97% complete agreement between all 3 abstractors (S.P., T.D. and S.R.) and a clinician (T.H.) experienced in melanoma for primary variables, including stage-defining items. Data on distant metastasis, which comprise the M-category, were supplemented by information on stage provided by regional cancer centres.
Classification of independent variables
Patient factors
Patient characteristics included age at diagnosis, sex and SES. Age and sex were ascertained from the OCR. SES was assigned using the Ontario Marginalization Index (ONMarg). The ON-Marg is the Ontario version of the Canadian Marginalization Index, an area-based socioeconomic measure developed to explore differences in marginalization between areas of Ontario.17 ON-Marg has previously been associated with health outcomes.17–19 The material deprivation dimension of the ON-Marg was used, incorporating such indicators as education, government subsidies and income.
Disease factors
Disease characteristics include histological subtype, anatomic location of the primary melanoma and ulceration status. Histological subtype and anatomic location were available in the OCR. Presence of ulceration was available from pathology abstraction. The presence of ulceration was used as a factor hypothesized to influence the ease of early detection of melanoma, affecting the strength of association between factors of interest and thickness of melanoma. This variable was thus tested for effect modification. When thickness was available but ulceration status was missing, ulceration was assigned as “absent.”
Health-system factors
Health region and rurality were investigated. Ontario is sub-divided into 14 health care regions called local health integration networks (LHINs), each responsible for funding, coordinating and providing health care services for their region.
Rurality was measured via the Rurality Index of Ontario. This index is a measure of the relative rurality of Ontario census subdivisions and measures geographic factors related to access to health services using a weighted formula that considers population size and density, travel time to the nearest basic referral centre and travel time to the nearest advanced referral centre.20,21 The Rurality Index of Ontario is based on a 0–100 scale, with higher scores indicating a greater degree of rurality.
Classification of dependent variable
The primary analyses were conducted with advanced melanoma defined as a Breslow thickness > 2.0 mm. Thickness was chosen given its strong independent prognostic value for overall survival and its relevance to most cases of melanoma diagnosed at the population level; advanced thickness is the most common reason for advanced stage, defined as American Joint Committee on Cancer (AJCC) stage II–IV. Unlike other stage-related variables (e.g., N- and M-category), thickness is available in pathology reports systematically collected by the OCR for the vast majority of patients.
Secondary analyses were conducted defining advanced melanoma as an AJCC 7th edition stage II and above. By definition, all melanomas > 2.0 mm are stage II or above. Data abstracted from pathology reports were used to derive AJCC stage. When elements of the AJCC stage were missing, minimum stage was assigned.
Statistical analyses
All statistical analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC). Univariate associations were assessed with χ2 statistics. All variables independently associated with advanced melanoma, with p < 0.20, were added into a mutually adjusted multivariable modified Poisson model with a robust error variance; variables remained in the model with p < 0.20. Effect modification was assessed by including interaction terms with ulceration status and each of the variables and assessing their significance. Sensitivity analyses were conducted to assess our assumptions regarding missing data. The Kaplan–Meier product-limit method was used to characterize survival stratified by the presence of advanced melanoma.
Ethics approval
This study was approved by the Queen’s University Health Sciences and Affiliated Teaching Hospitals Research Ethics Board (EPID-425-13).
Results
Study population
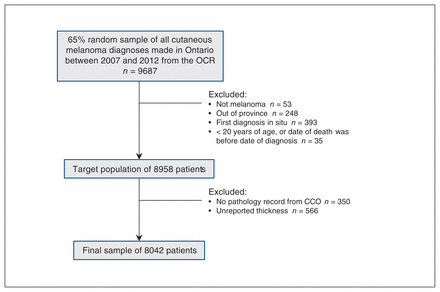
Our 65% random sample included 9687 patients with a diagnosis of cutaneous melanoma in the OCR between 2007 and 2012. Following exclusions, 8042 patients comprised our thickness-based study cohort (Figure 1).
Flow diagram for selection of thickness-based study cohort. Note: CCO = Cancer Care Ontario, OCR = Ontario Cancer Registry.
Table 1 presents cohort characteristics overall and stratified by ulceration. Table 2 presents the percentages of patients with each characteristic (patient, disease and health-system factors) who had advanced melanoma. There were significant differences for all variables (p < 0.001). Older patients, male patients and those living in the most deprived SES quintile were more likely to have advanced melanoma. Patients with nodular melanomas or with ulceration were also more likely to have advanced melanoma. Those with melanomas diagnosed on the head or neck and unspecified areas were more likely to have advanced disease.
Characteristics of the overall cohort with thickness information, and stratified by ulceration status
Presence of advanced melanoma (> 2.0 mm thickness) according to study factors, for the overall cohort and stratified by ulceration status
Effect modification
Interaction terms for the presence of ulceration were statistically significant (p < 0.05) for age, SES and histology, and body site approached significance (p = 0.05). For this reason, we performed analyses stratified by ulceration (Table 2 and Table 3). Similar significance of interactions was observed using the advanced AJCC stage definition (Appendix 1, available at www.cmajopen.ca/content/6/4/E502/suppl/DC1).
Relative risk of advanced melanoma from modified Poisson regression, including results stratified by ulceration status
Relative risk of advanced melanoma
Results for the univariate and multivariate modified Poisson regression are presented in Table 3. Univariate analyses revealed significant associations between all variables and advanced melanoma (p < 0.05). Notably, in bivariate analysis, associations between rurality and the variables for SES and LHIN were observed. When all variables were included in the modified Poisson model, rurality lost significance (p = 0.6), and it was removed from the final model.
After controlling for all variables in the final model, men had a 12% greater risk of being diagnosed with advanced melanoma than women (relative risk [RR] 1.12; 95% confidence interval [CI] 1.05–1.20). Risk of advanced melanoma also increased with age. For example, those between the ages of 76 and 85 years had a 27% greater risk than those aged 56 to 65 years (RR 1.27; 95% CI 1.16–1.40). In addition, patients living in neighbourhoods in the most deprived SES quintile had a 24% greater risk of advanced melanoma than those in the least deprived SES quintile (RR 1.24; 95% CI 1.12–1.38). There was also variation between the LHINs, with the RRs of individual LHINs ranging from 0.92 to 1.34 (p = 0.005). When stratified by ulceration status, disparities were greatest for non-ulcerated cases, and they were attenuated for many of the estimated RRs for ulcerated cases (Table 3); however, many remained significant. Similar, albeit attenuated, findings were observed using our definition of advanced melanoma based on AJCC stage when stratified by ulceration (Appendix 1).
Survival analyses
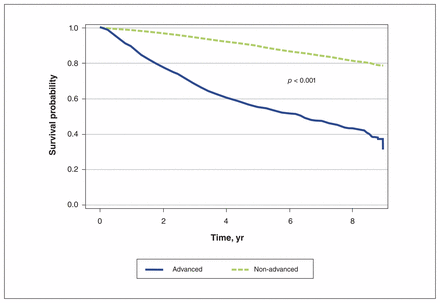
Five-year overall survival was 81% for our entire cohort. Survival of patients with advanced melanoma was 55.9%, compared with 89.7% for patients with non-advanced melanoma (p < 0.001; Figure 2).
Overall survival stratified by presence of advanced melanoma (> 2.0 mm thick). Five-year survival is 90% for patients with non-advanced melanoma and 56% for patients with advanced melanoma.
Sensitivity analyses
Several sensitivity analyses were conducted to ensure the robustness of the assumptions made for missing data. For cases with thickness data but missing ulceration data, there was a negligible difference in RRs in models with unreported ulceration set as “missing,” where unreported ulceration cases were excluded, and where unreported ulceration was set to “absent.”
It was hypothesized that patients with no pathology report more often had an advanced-stage cancer and were too ill for further testing. Indeed, those with no pathology reports had lower survival, had a higher proportion of melanoma not otherwise specified (NOS) in the OCR and were more likely to have an “unspecified” location of the primary melanoma than those who had a pathology report (data not shown). In a model assuming patients without pathology reports had an advanced-stage melanoma, there were negligible differences in RRs compared with the baseline model.
Interpretation
In this contemporary Canadian melanoma cohort from Ontario, we discovered substantial differences in risk of advanced melanoma for patients living in more deprived regions of the province and for patients living within certain health regions (LHINs). There was also a greater risk of advanced melanoma for men and for older patients. These findings are important given the large differences in survival observed for patients with advanced melanoma in our cohort. The disparities were greatest when ulceration was absent, which may hold relevance for the development and evaluation of early detection strategies for melanoma. These associations suggest that there may be inequitable access to timely diagnosis of cutaneous melanoma in Ontario, requiring further investigation and action.
There are important strengths to our study. Our cases of melanoma came from the OCR, which is population based. This provincial database is known for its level of population coverage, and a contemporary study suggests capture rates of 95.7% or more for melanoma in 2007–2009 even for community laboratory pathology reports.16,22 This coverage was important as melanoma can be diagnosed and treated in a variety of health care settings. As pathology reports for all cancer diagnoses are archived by the OCR, we could under-take primary data collection on pathologic stage information. This improved the generalizability of our findings to the population of Ontario and allowed us to characterize and measure the burden of advanced melanoma in Ontario using a population-based sample.
We found disparate rates of advanced melanoma according to sex, age, SES, LHIN, histology, ulceration and anatomic location. Our results suggest that each of these variables is independently associated with advanced melanoma in Ontario. Our stratified analyses suggest that larger disparities exist when ulceration is absent. To explain this finding, we hypothesize that disparities may be more pronounced when melanoma is asymptomatic (e.g., some non-ulcerated melanomas) or there is disparate awareness of certain warning signs between groups (e.g., the ABCDEs of melanoma: asymmetry, border, colour, diameter and evolution). The melanoma ABCDEs are perhaps the warning signs best known to the public, and together they are particularly relevant to superficial spreading melanoma; thick melanomas are more likely to be nodular, ulcerated, fast growing and non-pigmented.
Advanced age was associated with advanced melanoma. The reason is probably multifactorial. It may be that when melanoma is more difficult to detect, a situation captured indirectly in our study by lack of ulceration, older individuals are even less likely to self-detect a melanoma in its early stages or promptly seek medical attention than younger individuals. Other health issues and symptoms may be considered more pressing. It may also be that disparities in awareness of the early warning signs of melanoma exist by age and that this has a stronger influence on the detection of non-ulcerated melanomas.
After adjustment for other factors, men were still at a greater risk of being diagnosed with advanced melanoma than women (RR 1.12; 95% CI 1.05–1.20). There may be differences in health-seeking behaviour between the sexes or differences in tumour-related factors other than ulceration. In keeping with known epidemiology, men were more likely than women to be diagnosed with a trunk melanoma; trunk lesions on men most often occur on the back, impeding self-detection.23
We found variation in the risk of advanced melanoma according to an area-level measure of SES. Those in the lowest SES quintile (the most deprived) had a 24% increased risk of being diagnosed with advanced melanoma (RR 1.24; 95% CI 1.12–1.38). The relative risk of advanced melanoma was greater in this group when ulceration was absent. It may be that those living in more deprived neighbourhoods are less likely to appreciate the seriousness of their lesion until it displays more advanced features such as ulceration, or they may be unable to advocate for themselves when they suspect an unusual lesion.24 Moreover, there may be issues regarding access to care for those of lower SES. For instance, those of lower SES may be unable to afford travel to a specialist, particularly if they live outside of a major urban centre where specialists are concentrated. Notably, the number of dermatologists per 100 000 population is greatest in the Toronto Central, Central and Champlain LHINs, with a substantially lower supply of these specialists in all other LHINs.25
Finally, we observed variations in advanced melanoma diagnoses across health regions in Ontario, even after adjusting for other factors such as SES and age. Although the proportion of melanoma diagnoses closely followed the proportion of the population living in the health care region (data not shown), there was variation in the proportion of advanced melanoma diagnoses across the LHINs, ranging from 21% to 32% (p = 0.0009 across the LHINs). There may be system-level differences in access to care and/or quality of care. There is a need for research elucidating details of the diagnostic pathways and access to specialist care for patients in different LHINs. Variation in access to dermatologists and other skin care specialists across the LHINs is one hypothesis.
Limitations
There are several limitations to this study. There is a risk of misclassification of stage and pathologic prognostic factors. To mitigate this risk, thickness and stage data were collected directly from pathology reports using a standardized algorithm. We used ecologic measures of SES. Household and individual-level SES variables such as income can vary substantially within regions. This is a recognized limitation of any study using measures based on postal codes and is acknowledged. The lack of significance of rurality in our final model may have related to correlation with SES and LHIN. We did not investigate pure in situ disease as complete population data were unavailable. There were missing thickness data; however, our sensitivity analyses revealed that our assumptions were robust. A stage-based analysis was also undertaken. With some evidence of missingness not at random and limited missing data, multiple imputation was not employed. Finally, there is the possibility of residual confounding. For example, the presence of comorbidities has the potential to influence the association between several of our variables and advanced melanoma; however, comorbidity is correlated with age, sex and SES, which we controlled for in our analysis.26–28
Conclusion
This was a contemporary study of melanoma in a universal health care setting, adding to the limited population-level literature on the diagnosis of advanced melanoma in Canada. We discovered clinically relevant differences in the risk of advanced melanoma according to SES and health region (LHIN). There was also more advanced melanoma diagnosed in men and older individuals, which may relate in part to inequitable access to care, even within a universal health care setting. As expected, survival was substantially worse for patients with advanced melanoma in our cohort. Disparities were greater when ulceration was absent; this holds relevance for the development and evaluation of system-level interventions for early detection. Future research is required to delve into the reasons why these disparities in advanced melanoma diagnosis exist, to help improve early detection and potentially increase survival.
Acknowledgements
The authors thank Sarah Pickett, Tina Dyer, Sue Rohland and Will Langley for skillfully undertaking the pathology report screening and data abstraction for this study.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Meaghan Mavor, Harriet Richardson and Timothy Hanna designed the study. Meaghan Mavor, Harriet Richardson, Qun Miao and Timothy Hanna acquired and analyzed the data. All authors contributed to interpreting the data and writing the manuscript, approved the final version for publication and agreed to be accountable for all aspects of the work.
Funding: A studentship for Meaghan Mavor was funded by the Canadian Centre for Applied Research in Cancer Control (ARCC). ARCC receives core funding from the Canadian Cancer Society Research Institute (grant 2105-703549). Timothy Hanna holds a research chair and received pilot funding provided by the Ontario Institute for Cancer Research through funding provided by the Government of Ontario (IA-035). This study was supported by a Canadian Institutes of Health Research operating grant (MOP 137022).
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/6/4/E502/suppl/DC1.
Disclaimer: Parts of this material are based on data and information provided by Cancer Care Ontario. The opinions, results, view and conclusions reported in this paper are those of the authors and do not necessarily reflect those of Cancer Care Ontario. No endorsement by Cancer Care Ontario is intended or should be inferred.
References
- Copyright 2018, Joule Inc. or its licensors