Abstract
BACKGROUND: Very little direct evidence exists on use of corticosteroids in patients with coronavirus disease 2019 (COVID-19). Indirect evidence from related conditions must therefore inform inferences regarding benefits and harms. To support a guideline for managing COVID-19, we conducted systematic reviews examining the impact of corticosteroids in COVID-19 and related severe acute respiratory illnesses.
METHODS: We searched standard international and Chinese biomedical literature databases and prepublication sources for randomized controlled trials (RCTs) and observational studies comparing corticosteroids versus no corticosteroids in patients with COVID-19, severe acute respiratory syndrome (SARS) or Middle East respiratory syndrome (MERS). For acute respiratory distress syndrome (ARDS), influenza and community-acquired pneumonia (CAP), we updated the most recent rigorous systematic review. We conducted random-effects meta-analyses to pool relative risks and then used baseline risk in patients with COVID-19 to generate absolute effects.
RESULTS: In ARDS, according to 1 small cohort study in patients with COVID-19 and 7 RCTs in non–COVID-19 populations (risk ratio [RR] 0.72, 95% confidence interval [CI] 0.55 to 0.93, mean difference 17.3% fewer; low-quality evidence), corticosteroids may reduce mortality. In patients with severe COVID-19 but without ARDS, direct evidence from 2 observational studies provided very low-quality evidence of an increase in mortality with corticosteroids (hazard ratio [HR] 2.30, 95% CI 1.00 to 5.29, mean difference 11.9% more), as did observational data from influenza studies. Observational data from SARS and MERS studies provided very low-quality evidence of a small or no reduction in mortality. Randomized controlled trials in CAP suggest that corticosteroids may reduce mortality (RR 0.70, 95% CI 0.50 to 0.98, 3.1% lower; very low-quality evidence), and may increase hyperglycemia.
INTERPRETATION: Corticosteroids may reduce mortality for patients with COVID-19 and ARDS. For patients with severe COVID-19 but without ARDS, evidence regarding benefit from different bodies of evidence is inconsistent and of very low quality.
On Mar. 11, 2020, the World Health Organization declared coronavirus disease 2019 (COVID-19) a pandemic.1 The worldwide spread of COVID-19 represents a profound threat to human health.
Clinicians frequently treat patients with COVID-19 with corticosteroids. 2 Their use is controversial: 2 commentaries published recently in The Lancet expressed opposing views based partly on original studies of severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS) and influenza: 1 recommended against using corticosteroids, while the other recommended using corticosteroids in some patients with COVID-19.3,4
Formulating recommendations for clinicians regarding use of corticosteroids in patients with COVID-19 requires systematic summaries of the available evidence. Therefore, to support a clinical practice guideline addressing management of patients with COVID-19,5 we conducted a series of systematic reviews. Because we anticipated a paucity of direct evidence from patients with COVID-19, we included available evidence addressing corticosteroids in the treatment of acute respiratory distress syndrome (ARDS), SARS, MERS, influenza and community-acquired pneumonia (CAP), all providing indirect evidence that informs the efficacy and safety of corticosteroid use in patients with COVID-19.
Methods
For ARDS, we used definitions in eligible studies. For severe COVID-19, we used the World Health Organization definition of severity: fever or suspected respiratory infection, plus 1 of the following: respiratory rate > 30 breaths/min, severe respiratory distress, or peripheral oxygen saturation (SpO2) ≤ 93% on room air.6
For COVID-19, SARS and MERS, we conducted systematic reviews that sought all eligible primary studies. For ARDS, influenza and CAP, we chose the most recent methodologically rigorous systematic reviews and searched for recent eligible primary studies. Choice of outcomes were informed by our preliminary protocol, by guidance from the guideline panel, and from what authors of eligible studies reported.
Search strategies and selection criteria
Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) presents the protocol we developed before launching these systematic reviews, which follow the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).7
COVID-19, SARS and MERS
With the assistance of a medical librarian (R.J.C.), we searched MEDLINE, Embase, PubMed and the Cochrane Central Register of Controlled Trials from the date of their inception to Apr. 19, 2020, and searched medRxiv until Apr. 25, 2020. For studies of patients with COVID-19, we also searched Chinese databases, including China National Knowledge Infrastructure (CNKI), Wanfang, Chongqing VIP Information (CQVIP), and ChinaXiv. Appendix 2 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) presents the complete search strategy.
We included randomized controlled trials (RCTs), cohort and case–control studies comparing corticosteroids versus no corticosteroids in patients with COVID-19, SARS or MERS. For cohort studies and case–control studies, we included only studies that performed adjusted analysis unless all studies failed to conduct an adjusted analysis, in which case we included unadjusted analyses. For overlapping studies (studies that included patients from the same data sources), we included only the larger unless there was a specific additional helpful analysis in the smaller.
ARDS, influenza and CAP
We conducted separate searches for ARDS, influenza and CAP using a 2-stage process (for search strategy, see Appendix 2). First, to identify systematic reviews that examined the effect of corticosteroids on ARDS, influenza or CAP, we searched MEDLINE, Embase, the Cochrane Database of Systematic Reviews and Epistemonikos, and chose the most recent methodologically rigorous one. Second, we searched MEDLINE, Embase and ClinicalTrials.gov for ARDS and CAP, and searched MEDLINE, Embase, PubMed and the Cochrane Central Register of Controlled Trials for influenza, for studies published subsequent to the search of the chosen reviews. For ARDS and CAP, we included only RCTs. For influenza, we included RCTs and cohort studies.
For all searches, 2 reviewers independently screened titles and abstracts and, subsequently, full texts of potentially eligible studies to determine final eligibility. Disagreements were resolved by discussion or, if necessary, referral to a third reviewer. We applied no language restriction.
Data analysis
Two reviewers independently extracted study characteristics, with adjudication by a third reviewer if necessary. Outcomes included mortality, length of intensive care unit (ICU) stay, length of hospital stay, duration of mechanical ventilation, need for mechanical ventilation, viral ribonucleic acid (RNA) clearance, viral shedding time, serious hyperglycemia, superinfection, neuromuscular weakness and gastrointestinal bleeding.
We calculated summary estimates using Stata or Review Manager and calculated relative effects (odds ratios [ORs], risk ratios [RRs] or hazard ratios [HRs]) and 95% confidence intervals (95% CIs) for dichotomous outcomes, and mean differences (MDs) and 95% CIs for continuous outcomes using a random-effects model. For continuous outcomes and adjusted estimates, we used the inverse variance ( DerSimonian and Laird) method; for dichotomous outcomes from RCTs, we used the Mantel–Haenszel method. We assessed inconsistency among studies by differences in point estimates and overlap of the confidence intervals, and the I2 statistic. For dichotomous outcomes, we calculated the absolute treatment effects by applying relative effects to risk in patients not receiving corticosteroids in 2 groups: patients with severe COVID-19 and patients with COVID-19 and ARDS. We chose the baseline mortality risk of patients with COVID-19 and ARDS from an observational study of patients with COVID-19 and ARDS,8 and the baseline mortality risk of patients with severe COVID- 19 from an observational study of patients with severe COVID-19.2 For other outcomes, we relied for baseline risks on the medians of the groups not receiving corticosteroids in the included studies.
Risk of bias assessment
We used the ROBIS risk of bias tool9 to choose the most methodologically rigorous systematic review to be updated. We used a modified version of the Cochrane risk of bias tool10 to assess risk of bias in RCTs, and a revised version of the Newcastle–Ottawa Scale11,12 for observational studies (details available at www.evidencepartners.com/resources/methodological-resources/). Two reviewers independently assessed risk of bias, resolving disagreements with a third reviewer if necessary.
Rating of evidence quality
We used the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach to rate the quality of evidence for each outcome as high, moderate, low or very low.13 The assessment included judgments addressing risk of bias,14 imprecision, 15 inconsistency,16 indirectness17 and publication bias.18 If there were serious concerns in any of these domains (for instance, in risk of bias), we rated down the quality of the evidence. Because the effect of corticosteroids in these diseases might differ from effects in the COVID-19 population, using the GRADE approach, for benefit outcomes in SARS and MERS, we rated down 1 level for indirectness, and for ARDS, influenza and CAP, we rated down 2 levels. Because we considered estimates of harm to be more likely to apply across populations than benefit outcomes, for all populations we rated down 1 level for harms.
Ethics approval
Ethics approval was not required for this systematic review.
Results
Appendix 3 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) presents the study selection process. Our search for COVID-19, SARS and MERS identified 5120 citations. After removing duplicates, screening titles and abstracts, and reviewing full texts, we ultimately included 1 cohort study8 including 84 patients with COVID-19 and ARDS, 5 cohort studies19–23 including 679 patients with COVID-19 but without ARDS, 3 studies (2 cohort studies24,25 and 1 RCT26) including 7087 patients with SARS, and 2 cohort studies27,28 including 623 patients with MERS.
Our search for systematic reviews of ARDS identified 836 citations; we ultimately chose a systematic review published in 2019 as the target for updating.29 Our search for primary studies identified 1 new eligible RCT published in 2020.30 Including 6 RCTs identified from the previous review, we included 7 RCTs30–36 with 851 patients.
Our search for systematic reviews for influenza identified 525 citations; we ultimately chose a systematic review published in 2019 as the target for updating.37 Our search for primary studies identified 1 new eligible study published in 2020.38 Including 30 studies identified from the previous review, we identified 31 eligible studies,39–69 of which 21 with 9536 patients were included in meta-analyses.41,43–47,50,52,53,55–61,63–65,68,69
Our search for systematic reviews for CAP identified 346 citations. We ultimately chose a systematic review published in 2015 as the target for updating.70 Our search for primary studies identified 1 new eligible study published in 2016.71 With 12 RCTs from the previous review, our systematic review included 13 RCTs71–83 including 2095 patients.
Appendix 4 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) presents characteristics of the included studies. Appendix 5 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) presents the risk of bias assessment for each study. Forest plots of the meta-analysis results are shown in Figures 1–⇓⇓⇓5 for mortality and in Appendix 6 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.200645/-/DC1) for other outcomes.
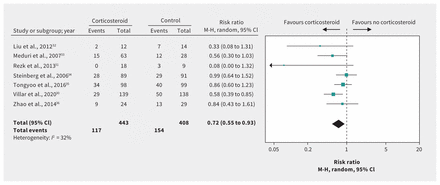
Effect of corticosteroids on mortality in patients with acute respiratory distress syndrome without coronavirus disease 2019. Note: CI = confidence interval, M-H = Mantel–Haenszel.
Effect of corticosteroids on mortality in patients with severe coronavirus disease 2019. Weights are from random-effects analysis. Note: CI = confidence interval, HR = hazard ratio, IV = inverse variance.
Effect of corticosteroids on mortality in patients with severe acute respiratory syndrome. Weights are from random-effects analysis. Note: CI = confidence interval, HR = hazard ratio, IV = inverse variance.
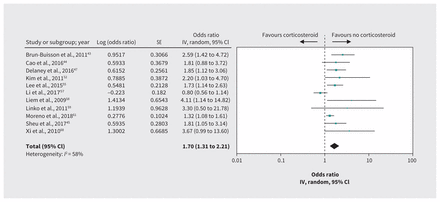
Effect of corticosteroids on mortality in patients with influenza. Note: CI = confidence interval, IV = inverse variance, SE = standard error.
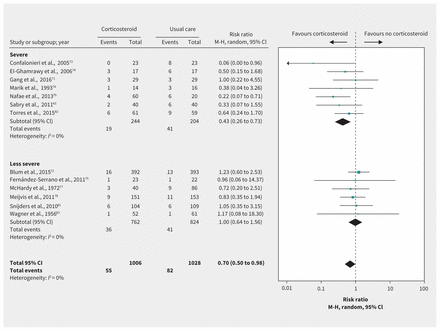
Effect of corticosteroids on mortality in patients with community-acquired pneumonia. Note: CI = confidence interval, M-H = Mantel–Haenszel.
ARDS
Evidence for patients with COVID-19 and ARDS was available from a single observational study of 84 patients8 that suggested corticosteroids may result in a large mortality reduction compared with no corticosteroids (HR 0.41, 95% CI 0.20 to 0.83, MD 29.2% lower; very low-quality evidence) (Table 1).
GRADE summary of findings: corticosteroids in patients with COVID-19 and ARDS, based on direct evidence from observational studies of patients with COVID-19 and ARDS
Evidence for ARDS without COVID-19 was available from 7 RCTs30–36 including 851 patients (Table 2). We considered the evidence for most outcomes to be high quality for patients with ARDS in general. After rating down 2 levels for indirectness of populations, we considered the evidence to be low quality for COVID-19. These RCTs suggest that corticosteroids may substantially reduce mortality (RR 0.72, 95% CI 0.55 to 0.93, MD 17.3% lower; low-quality evidence) (Figure 1). Very low-quality evidence raised the possibility that corticosteroids may have little or no impact on length of ICU stay32–34 (MD 0.1 days longer, 95% CI 3.0 days shorter to 3.2 days longer) but may reduce length of hospital stay33,34,36 (MD 3.6 days shorter, 95% CI 0.02 to 7.2 days shorter). Low-quality evidence shows that corticosteroids may reduce the duration of mechanical ventilation (MD −4.8 days, 95% CI −7.0 to −2.6),30,31,33–36 but increase serious hyperglycemia (risk increase 8.1%, 95% CI 0.7% to 16.2%),30,33,35 with few or no adverse effects on neuromuscular weakness, 33,34 gastrointestinal bleeding35,36 and superinfection.30,33–36
GRADE summary of findings: corticosteroids in patients with COVID-19 and ARDS, based on indirect evidence from randomized controlled trials of patients with ARDS but without COVID-19
Severe COVID-19: direct evidence from observational studies
Very low-quality evidence from 2 cohort studies19,23 including 331 patients with severe COVID-19 raised the possibility that corticosteroids may increase mortality compared with no corticosteroids (HR 2.30, 95% CI 1.00 to 5.29, MD 11.9% more) (Table 3, Figure 2). One cohort study20 reported an increase in the composite outcome of mortality or ICU admission with steroid use. Two cohort studies21,22 suggested that corticosteroids use was associated with prolonged viral shedding (very low-quality evidence).
GRADE summary of findings: corticosteroids in patients with severe COVID-19, based on direct evidence from observational studies of patients with severe COVID-19
Severe COVID-19: indirect evidence from observational studies and a randomized trial of SARS
Two cohort studies24,25 including 6129 patients with SARS provide low-quality evidence for corticosteroid impact on mortality in these patients, with additional consideration of indirectness in serious COVID-19 pneumonia (HR 0.83, 95% CI 0.41 to 1.66; very low-quality evidence) (Table 4, Figure 3). An RCT26 in which 16 patients with SARS treated with ribavirin were randomized to corticosteroids or no corticosteroids raised the possibility that early (< 7 days of illness) hydrocortisone therapy may increase the median time for SARS-associated coronavirus (SARS-CoV) RNA to become undetectable in plasma (MD 4.0 days longer, 95% CI 2.0–6.0 days; very low-quality evidence for SARS with additional consideration of indirectness in COVID-19) (Table 4).
GRADE summary of findings: corticosteroids in patients with severe COVID-19, based on indirect evidence from randomized controlled trials and observational studies of patients admitted to hospital with SARS
Severe COVID-19: indirect evidence from observational studies of MERS
One cohort study28 that enrolled 290 patients with MERS suggests a possible reduction in mortality with administration of corticosteroids (OR 0.75, 95% CI 0.52 to 1.07; very low-quality evidence for MERS with additional consideration of indirectness in COVID- 19) (Table 5). Data from 189 patients in the same study28 suggest that corticosteroid use may be associated with a delay in Middle East respiratory syndrome coronavirus (MERS-CoV) RNA clearance (HR 0.35, 95% 0.17 to 0.72; very low-quality evidence for MERS with additional consideration of indirectness for COVID-19) (Table 5).
GRADE summary of findings: corticosteroids in patients with severe COVID-19, based on indirect evidence from observational studies of patients admitted to hospital with MERS
Severe COVID-19: indirect evidence from observational studies of influenza
Evidence in patients with influenza from 11 cohort studies43–45,47,52,55,57–59,61,68 including 8530 patients with adjusted effect estimates for mortality suggests that corticosteroids may increase mortality (OR 1.70, 95% CI 1.31 to 2.21, MD 6.1% higher; low-quality evidence for influenza rated down to very low for indirectness) (Table 6, Figure 4). Very low-quality evidence for influenza with additional consideration of indirectness when applied to COVID-19 from cohort studies that failed to conduct an adjusted analysis raised the possibility that corticosteroids may increase the rate of superinfection (OR 2.74, 95% CI 1.51 to 4.95)43,44,47,52,55,57,65 and increase the number of patients requiring mechanical ventilation (OR 5.54, 95% CI 1.83 to 16.80)52,57,59,61 (Table 6).
GRADE summary of findings: corticosteroids in patients with severe COVID-19, based on indirect evidence from observational studies of patients admitted to hospital with influenza
Severe COVID-19: indirect evidence from randomized trials of CAP
Thirteen RCTs71–83 including 2034 patients with CAP addressed a number of important efficacy outcomes. For patients with CAP in general, evidence varied from high to low quality. After we rated down 2 levels for indirectness, all evidence for these outcomes was of low or very low quality. Corticosteroids were associated with reductions in mortality (RR 0.70, 95% CI 0.50 to 0.98, MD 3.1% lower), need for mechanical ventilation72,75,76,79,82 (risk difference [RD] 10.4%, 95% CI 4.3% to 13.8%), duration of mechanical ventilation71,73,74,79,80 (MD 3.5 days shorter, 95% CI 1.8 to 5.2 days), length of ICU stay;72–76,78,79,82 and length of hospital stay71–76,78,79,81,82,84 (Table 7, Figure 5). Meta-analysis of 8 RCTs71,72,75,78,79,81,82,84 showed that corticosteroids may increase the rate of serious hyperglycemia (RD 5.7%, 95% CI 0.18% to 15.3%; moderate-quality evidence for CAP, low quality after rating down 1 level for indirectness).
GRADE summary of findings: corticosteroids in patients with severe COVID-19, based on indirect evidence from randomized controlled trials of patients admitted to hospital with community-acquired pneumonia
Mortality results suggested a possible subgroup effect of corticosteroids by pneumonia severity (severe pneumonia, RR 0.43, 95% CI 0.26 to 0.73; less severe pneumonia, RR 1.00, 95% CI 0.64 to 1.56; p for interaction 0.02). However, the apparent effect is based on differences between rather than within studies, is driven to a considerable extent by a small study73 that was stopped early for benefit, almost certainly represents a large overestimate of effect, and does not appear with any other outcome. Thus, the subgroup effect has low credibility.
For other adverse events (neuropsychiatric events;72,81,82,84 superinfection71–74,78,81,82,84 and gastrointestinal bleeding71–75,79,80,82), evidence was moderate quality for small, no, or uncertain harms of corticosteroids in patients with CAP, and low quality after rating down once for indirectness (Table 7).
Interpretation
This series of systematic reviews informed a guideline addressing management of patients with COVID-19.5 Direct evidence from 1 observational study8 of 84 patients with COVID-19 and ARDS was consistent with the findings of our systematic review of RCTs of patients without COVID-19 that suggested corticosteroids may reduce mortality in patients with COVID-19 and ARDS by more than 15% and reduce the duration of mechanical ventilation. The evidence suggested corticosteroids may increase the rate of serious hyperglycemia, although not of other potentially worrisome adverse effects. The evidence for these effects is mostly of low quality.
For patients who have severe COVID-19 but are not critically ill, direct evidence from observational studies provided very low-quality evidence of an increase in mortality with corticosteroids. In SARS and MERS, evidence from observational studies raises the possibility of a modest mortality reduction with corticosteroids, but also of a delay in viral clearance. In CAP, RCT evidence also raises the possibility of a mortality reduction with corticosteroids and other benefits including reduction in length of hospital and ICU stay, and need for and duration of mechanical ventilation. Low-quality evidence suggests a likely increase in hyperglycemia and possible small increases in neuropsychiatric events and superinfection, but not in gastrointestinal bleeding. Observational studies in influenza provide discrepant findings, raising the possibility of substantial increases in mortality, superinfection and mechanical ventilation with corticosteroids.
Strengths of this review include a comprehensive search, independent study selection, data abstraction and risk of bias assessment by 2 reviewers and presentation of absolute effects for dichotomous outcomes. We rated the quality of evidence with the GRADE approach, paying close attention to important methodological issues such as differences in the impact of indirectness of evidence on benefit and harm outcomes. We are more skeptical of making inferences regarding benefits in patients with COVID-19 from other patient populations than we are of making inferences on harms. For observational studies, we included, as far as possible, only those with adjusted analyses. Finally, a particular strength is the presentation of a comprehensive assessment of all the indirect evidence, including from ARDS, SARS, MERS, influenza and CAP, together in a single document.
We compared our review with another published systematic review addressing corticosteroid therapy in COVID-19.85 Apart from COVID-19, SARS and MERS, our review included 3 additional populations: ARDS, CAP and influenza. We updated our search until Apr. 19, including evidence published more recently than the previous systematic review, which searched until Mar. 15.8,19–23 Third, we included, as far as possible, only cohort and case–control studies with adjusted effect estimates. Finally, we used GRADE to rate the quality of evidence.
For ARDS, our review showed similar results to the 1 other published systematic review29 that included the latest published studies. For CAP, the results on which we focus are similar to those of other recent reviews86–89 that showed that corticosteroids may reduce mortality and length of hospital stay, and increase hyperglycemia.
The findings for influenza are consistent with other previous systematic reviews90–92 that also found increased mortality associated with corticosteroid use. One review90 focused on patients with influenza pneumonia only, excluding those with mild illness or those in the ICU. The results showed that corticosteroids were associated with higher mortality. In contrast, another review92 studied severe forms of influenza and reported that among studies with adjusted estimates, results showed no statistically significant difference between the corticosteroid and control groups.
Limitations
The limitations of this study are largely those of the underlying evidence, which is either of low or, for benefits, very low quality for the most part. One could argue that we should have broadened our consideration of indirect evidence. For instance, we could have included Pneumocystis jiroveci pneumonia, in which evidence supports corticosteroid use. Our threshold was based on patients with viral pneumonia being included in the population, which is clearly the case for SARS, MERS and influenza, but also true for ARDS and CAP.
Similarly, with respect to harms, consideration of evidence from RCTs of short-term use of corticosteroids in other conditions might have strengthened our findings. We have, however, moderate-quality evidence in patients with ARDS of no important increase in superinfection, and low-quality evidence of an increase in serious hyperglycemia. Low-quality evidence suggests a possible small increase in neuropsychiatric events. For this outcome, evidence from other conditions might have been particularly helpful.
Conclusion
Given the paucity of direct evidence and the limitations of indirect evidence, it is critical for clinicians and researchers to cooperate in conducting high-quality studies, in particular large and rigorous RCTs, to evaluate the effect of corticosteroids in both patients with COVID-19 and ARDS and patients with severe COVID-19 but who are not critically ill. Fortunately, RCTs, including those that address corticosteroid treatment, are ongoing.
Acknowledgements
Quazi Ibrahim, Lehana Thabane, Diane Heels- Ansdell, Jason Busse and Li Wang provided support and suggestions for the statistical analysis. Yingqi Xiao is supported by the China Scholarship Council.
Footnotes
Competing interests: Bram Rochwerg is an investigator in a trial, supported by a Canadian Institute of Health Research grant, evaluating the effect of corticosteroids in COVID-19 patients. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Zhikang Ye and Gordon Guyatt contributed to the conception of the work. Zhikang Ye, Ying Wang, Luis Enrique Colunga-Lozano, Manya Prasad and Gordon Guyatt contributed to the design of the work. Rachel Couban, Zhikang Ye, Ying Wang, Luis Enrique Colunga-Lozano, Manya Prasad, Wimonchat Tangamornsuksan, Liang Yao, Shahrzad Motaghi, Maryam Ghadimi, Malgorzata Bala, Huda Gomaa, Fang Fang and Yingqi Xiao contributed to the acquisition, analysis and interpretation of data. Zhikang Ye, Ying Wang, Luis Enrique Colunga-Lozano and Manya Prasad drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work. Zhikang Ye, Ying Wang, Luis Enrique Colunga-Lozano and Manya Prasad are joint primary authors.
Funding: None.
Data sharing: Data extracted from the included studies are presented in the results; however, full extraction data tables are available upon reasonable request from the corresponding author.
- Accepted May 1, 2020.