Abstract
Background
Characterization of breast cancer phenotypes has improved our ability to predict breast cancer behavior. Triple-negative (TN) breast cancers have higher and earlier rates of distant events. It has been suggested that this behavior necessitates treating TNs faster than others, including use of neoadjuvant chemotherapy (NACT) if time to surgery is not rapid.
Methods
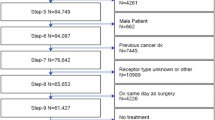
A review of women diagnosed with non-inflammatory, invasive breast cancer was conducted using the National Cancer Database for patients not having NACT, diagnosed between 2010 and 2014. Changes in overall survival due to delay were measured by phenotype.
Results
Overall, 351,087 patients met the inclusion criteria, including 36,505 (10.4%) TNs, 77.9% hormone receptor-positive (HR+) and 11.7% human epidermal growth factor receptor 2 (HER2)-enriched (HER2+). Phenotype, among other factors, was predictive of treatment delays. Adjusted median days from diagnosis to surgery and chemotherapy were 29.9, 31.6 and 31.5 (p< 0.001), and 72.7, 78.0 and 74.4 (p< 0.001) for TNs, HR+ and HER2+ cancers, respectively. After diagnosis, OS declined for all patients per month of preoperative delay (hazard ratio 1.104; p< 0.001). In models separating or combining surgery and chemotherapy, this survival decline did not vary by breast cancer phenotype (p > 0.3).
Conclusions
Delays cause small but measurable effects overall, but the effect on survival does not differ among breast cancer phenotypes. Our data suggest that urgency between diagnosis and surgery or chemotherapy is similar for breast cancers of different subtypes. Although NACT is sometimes advocated solely to avoid treatment delays, this study does not suggest a greater surgical urgency for TNs compared with other breast cancer phenotypes.

Similar content being viewed by others
References
Bleicher RJ, Ruth K, Sigurdson ER, et al. Trends in the patient’s preoperative time burden during the evaluation of breast cancer. Oncology 2013;27(Suppl 1):22–23.
Dinan MA, Curtis LH, Hammill BG, et al. Changes in the use and costs of diagnostic imaging among Medicare beneficiaries with cancer, 1999–2006. JAMA. 2010;303:1625–31.
Hillen MA, Medendorp NM, Daams JG, et al. Patient-driven second opinions in oncology: a systematic review. Oncologist. 2017;22:1197–1211.
Churilla TM, Egleston BL, Murphy CT, et al. Patterns of multidisciplinary care in the management of non-metastatic invasive breast cancer in the United States Medicare patient. Breast Cancer Res Treat. 2016;160:153–62.
Bleicher RJ, Ruth K, Sigurdson ER, et al. Preoperative delays in the US medicare population with breast cancer. J Clin Oncol. 2012;30:4485–92.
Polverini AC, Nelson RA, Marcinkowski E, et al. Time to treatment: measuring quality breast cancer care. Ann Surg Oncol. 2016;23:3392–402.
Bleicher RJ, Ruth K, Sigurdson ER, et al. Time to surgery and breast cancer survival in the United States. JAMA Oncol. 2016;2:330–9.
Parker JS, Mullins M, Cheang MCU, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27:1160–67.
Kumar P, Aggarwal R. An overview of triple-negative breast cancer. Arch Gynecol Obstet. 2016;293:247–69.
Chavez-MacGregor M, Clarke CA, Lichtensztajn DY, et al. Delayed initiation of adjuvant chemotherapy among patients with breast cancer. JAMA Oncol. 2016;2:322–9.
Bilimoria KY, Stewart AK, Winchester DP, et al. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15:683–90.
Bleicher RJ. Timing and delays in breast cancer evaluation and treatment. Ann Surg Oncol. 2018;25:2829–38.
Liederbach E, Sisco M, Wang C, et al. Wait times for breast surgical operations, 2003–2011: a report from the National Cancer Data Base. Ann Surg Oncol. 2015;22:899–907.
Smith EC, Ziogas A, Anton-Culver H. Delay in surgical treatment and survival after breast cancer diagnosis in young women by race/ethnicity. JAMA Surg. 2013;148:516–23.
Bleicher RJ, Chang C, Wang CE, et al. Treatment delays from transfers of care and their impact on breast cancer quality measures. Breast Cancer Res Treat. 2019;173:603–17.
Chia SK, Bramwell VH, Tu D, et al. A 50-gene intrinsic subtype classifier for prognosis and prediction of benefit from adjuvant tamoxifen. Clin Cancer Res. 2012;18:4465–72.
Carey LA, Dees EC, Sawyer L, et al. The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res. 2007;13:2329–34.
Prat A, Pineda E, Adamo B, et al. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast. 2015;24(Suppl 2):S26–35.
Yagata H, Kajiura Y, Yamauchi H. Current strategy for triple-negative breast cancer: appropriate combination of surgery, radiation, and chemotherapy. Breast Cancer. 2011;18:165–73.
Eastman A, Tammaro Y, Moldrem A, et al. Outcomes of delays in time to treatment in triple negative breast cancer. Ann Surg Oncol. 2013;20:1880–5.
Morante Z, De la Cruz-Ku G, Pinto J, et al. Localized triple negative breast cancer in extremes of life [abstract no. P1-15-06]. In: Proceedings of the 2017 San Antonio Breast Cancer Symposium. Cancer Res. 2018;78.
Desch CE, McNiff KK, Schneider EC, et al. American Society of Clinical Oncology/National Comprehensive Cancer Network Quality Measures. J Clin Oncol. 2008;26:3631–7.
Gagliato Dde M, Gonzalez-Angulo AM, Lei X, et al. Clinical impact of delaying initiation of adjuvant chemotherapy in patients with breast cancer. J Clin Oncol. 2014;32:735–44.
Silber JH, Rosenbaum PR, Clark AS, et al. Characteristics associated with differences in survival among black and white women with breast cancer. JAMA. 2013;310:389–97.
Rastogi P, Anderson SJ, Bear HD, et al. Preoperative chemotherapy: updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27. J Clin Oncol. 2008;26:778–85.
Acknowledgments
This work was supported by a United States Public Health Services grant for analysis of the data via support of our biostatistics facility, and by generous private donor support from the Marlyn Fein Chapter of the Fox Chase Cancer Center Board of Associates, for analysis and interpretation of the data. The NCDB is a joint project of the CoC of the ACS and the American Cancer Society. The data used in the study are derived from a de-identified NCDB file. The ACS and the CoC have not verified and are not responsible for the analytic or statistical methodology employed, or the conclusions drawn from these data by the investigator.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Alina M. Mateo, Anna M. Mazor, Elias Obeid, John M. Daly, Elin R. Sigurdson, Elizabeth A. Handorf, Lyudmila DeMora, Allison A. Aggon, and Richard J. Bleicher declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mateo, A.M., Mazor, A.M., Obeid, E. et al. Time to Surgery and the Impact of Delay in the Non-Neoadjuvant Setting on Triple-Negative Breast Cancers and Other Phenotypes. Ann Surg Oncol 27, 1679–1692 (2020). https://doi.org/10.1245/s10434-019-08050-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-08050-y