Abstract
Background: Characterizing the multiorgan manifestations and outcomes of patients hospitalized with COVID-19 will inform resource requirements to address the long-term burden of this disease. We conducted a descriptive analysis using prospectively collected data to describe the clinical characteristics and spectrum of organ dysfunction, and in-hospital and longer-term clinical outcomes of patients hospitalized with COVID-19 during the first wave of the pandemic at a Canadian centre.
Methods: We conducted a prospective case series involving adult patients (aged ≥ 18 yr) with COVID-19 admitted to 1 of 2 hospitals in London, Ontario, from Mar. 17 to June 18, 2020, during the first wave of the pandemic. We recorded patients’ baseline characteristics, physiologic parameters, measures of organ function and therapies administered during hospitalization among patients in the intensive care unit (ICU) and in non-ICU settings, and compared the characteristics of hospital survivors and nonsurvivors. Finally, we recorded follow-up thoracic computed tomography (CT) and echocardiographic findings after hospital discharge.
Results: We enrolled 100 consecutive patients (47 women) hospitalized with COVID-19, including 32 patients who received ICU care and 68 who received treatment in non-ICU settings. Respiratory sequelae were common: 23.0% received high-flow oxygen by nasal cannula, 9.0% received noninvasive ventilation, 24.0% received invasive mechanical ventilation and 2.0% received venovenous extracorporeal membrane oxygenation. Overall, 9.0% of patients had cerebrovascular events (3.0% ischemic stroke, 6.0% intracranial hemorrhage), and 6.0% had pulmonary embolism. After discharge, 11 of 19 patients had persistent abnormalities on CT thorax, and 6 of 15 had persistent cardiac dysfunction on echocardiography.
Interpretation: This study provides further evidence that COVID-19 is a multisystem disease involving neurologic, cardiac and thrombotic dysfunction, without evidence of hepatic dysfunction. Patients have persistent organ dysfunction after hospital discharge, underscoring the need for research on long-term outcomes of COVID-19 survivors.
The typical clinical spectrum of COVID-19, the illness caused by SARS-CoV-2, ranges from mild respiratory symptoms to multiorgan failure and death.1,2 Emerging evidence has shown that COVID-19 is associated with a range of pulmonary and extrapulmonary organ involvement.3 Although early Canadian data shed light on the outcomes and mortality rate, studies on the morbidity of hospitalized patients have been sparse and often based on retrospectively collected data,4–6 and many have excluded patients hospitalized outside the intensive care unit (ICU).5–7
To characterize the clinical course, multiorgan involvement and outcomes of COVID-19, prospectively collected data are required. Such data may potentially inform Canadian health care priorities as they relate to both the long-term burden of COVID-19 and future pandemics. We conducted a descriptive analysis using prospectively collected data to describe the clinical characteristics and spectrum of organ dysfunction, and in-hospital and longer-term clinical outcomes of hospitalized patients with COVID-19 during the first wave of the pandemic at a Canadian centre.
Methods
Study design and setting
This prospective case series was conducted at 2 sites in London Health Sciences Centre, a 1116-bed academic, tertiary care centre in London, Ontario, that comprises 2 hospitals: Victoria Hospital and University Hospital. We report this study in compliance with Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies.8
Patient recruitment
We consecutively enrolled all adult patients admitted to hospital with a diagnosis of COVID-19 from Mar. 17 to June 18, 2020. Inclusion criteria included symptoms consistent with COVID-19,1 age 18 years or older and a diagnosis of COVID-19 confirmed by a positive nasopharyngeal or tracheal aspirate polymerase chain reaction assay, either 7 days before hospital admission (not including the day of hospital admission) or at the time of the index hospitalization. We excluded patients who declined to participate in the study.
Data collection
Several study authors (D.C., K.D., K.F., D.G., K.H. and J.B.) screened all hospital admissions on a daily basis, enrolled eligible patients, and collected data prospectively using a pilottested case report form on the Research Electronic Data Capture (REDCap) platform, hosted by the Lawson Health Research Institute. We employed branching logic and numerical validations in the REDCap case report form to enhance data accuracy. One of the principal investigators (J.B. or K.H.) reviewed all data collected daily to ensure accuracy and completeness.
We recorded patients’ baseline demographic characteristics, physiologic parameters, investigations (including incidence and findings of computed tomography [CT], echocardiography and lung ultrasonography studies), therapies administered throughout hospitalization in non-ICU and ICU settings, and overall outcome (i.e., in-hospital death v. discharge from hospital and discharge destination). Finally, we recorded any echocardiography and thoracic CT scans performed after hospital discharge for clinical indications up until Dec. 31, 2020.
Echocardiography was conducted by an echocardiography technologist and the data were extracted by one of the study authors (J.B.), who is an intensive care physician with advanced training in point-of-care ultrasonography. Lung ultrasound images were acquired and reported by emergency physicians and critical care physicians. Data extraction was done by a study author (J.B.). Computed tomography reports were dictated by a radiologist. Data from the final radiology report were extracted by one of the study investigators. We collected data from patients and their clinical teams, paper charts, electronic medical records and the Critical Care Information System database. Appendix 1 (available at www.cmajopen.ca/content/10/3/E675/suppl/DC1) provides a description of all data elements and sources.
To ensure that our coding of the reports was relevant and comprehensive, we used the expertise of the study team. The study team consists of experts in these areas, including a respirologist (M.J.N.) and 2 intensive care physicians with advanced training and expertise in point-of-care lung ultrasonography and echocardiography (J.B. and R.A.), one of whom (R.A.) is an internationally recognized expert in advanced point-of-care echocardiography. This expertise facilitated the coding of relevant CT chest and echocardiographic data to ensure that we recorded relevant information in a consistent and accurate way, especially since we aimed to use the data to delineate derangements in cardiac physiology as a result of COVID-19.
Statistical analysis
We used descriptive statistics (means and standard deviations, medians and interquartile ranges [IQRs], and proportions and percentages, as appropriate) to summarize patients’ baseline characteristics, physiologic parameters, therapies and outcomes.
We applied the Wilcoxon rank-sum test for non-normally distributed continuous variables and the χ2 statistic for categorical variables, as appropriate, to compare characteristics of hospital survivors with those of nonsurvivors. We also compared lung ultrasound findings of patients who were not mechanically ventilated with the findings of those who were mechanically ventilated, expressed as an odds ratio with 95% confidence interval. In addition, we compared the characteristics of patients who were admitted to and remained in a non-ICU location with characteristics of patients who were initially admitted to a non-ICU location but were subsequently transferred to ICU because of clinical deterioration.
We selected variables on the basis of prior retrospective data showing an association between the variable and outcomes of interest.1,9 They included age, vital signs at emergency department triage, and laboratory data obtained on admission, such as blood pH, white blood cell count, hemoglobin, platelets, lymphocyte count, lactate, ferritin, lactate dehydrogenase, C-reactive protein and troponin levels. All statistical tests are 2-sided, with the threshold of statistical significance set at a p value of less than 0.05. We performed all data analyses using SPSS version 25.0 (IBM).
Ethics approval
Western University’s Research Ethics Board approved the conduct of this study (study no. 115732; Apr. 1, 2020).
Results
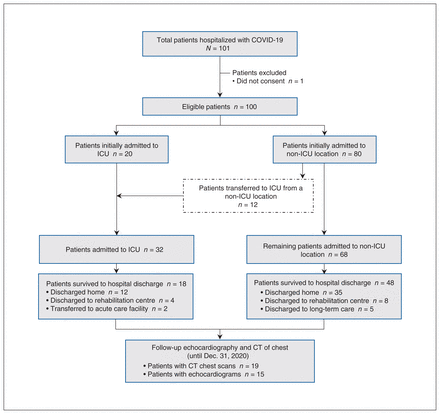
From Mar. 17 to June 18, 2020, 101 patients were admitted to 1 of the participating hospitals with a positive SARS-CoV-2 test. Of those, 1 patient declined to participate in the study. We included 100 patients (32 ICU and 68 non-ICU patients). Figure 1 provides an overview of the recruitment procedures and patients’ clinical disposition in hospital. Table 1 presents patients’ baseline characteristics.
Overview of patient enrolment and disposition. Note: CT = computed tomography, ICU = intensive care unit.
Baseline characteristics of patients admitted to hospital with COVID-19
Clinical presentation
Table 2 presents patients’ clinical characteristics at the time of hospital admission. Symptoms reported by more than 50% of patients at the time of hospitalization for COVID-19 were cough, dyspnea, fever and fatigue. At the time of assessment in the emergency department, 44.2% (42/95) of patients presented with a respiratory rate greater than 24 breaths/min, and 35.4% (35/99) received supplemental oxygen therapy in the emergency department to maintain oxygen saturation levels above 92%.
Clinical characteristics at the time of presentation to hospital
Twenty patients were admitted to the ICU, and 80 patients were admitted to the ward. Among the 80 patients initially admitted to the ward, 12 were eventually transferred to the ICU after a median hospital stay of 0.5 (IQR 0–2) days. Ultimately, 32 patients were admitted to the ICU.
Complications and management by organ system
Respiratory complications and therapies
Table 3 shows the respiratory complications and therapies received during hospitalization. Among all 100 patients, 79 (79.0%) received supplemental oxygen therapy, including high-flow nasal cannula (23/100), noninvasive ventilation (9/100), invasive mechanical ventilation (24/100) and venovenous extracorporeal membrane oxygenation (2/100). Prone positioning included awake self-proning in non-ICU patients (4/68 patients) and standard proning of ventilated patients in the ICU (13/30). Most ICU patients (24/32) received invasive mechanical ventilation, with a median duration of 14 (IQR 10–22) days.
Pulmonary complications and management among patients hospitalized with COVID-19
Twenty-six patients underwent CT of the thorax. The most common findings were bilateral diffuse ground-glass opacities, bilateral consolidations and new fibrosis or bronchiectasis (Table 3). Lung ultrasonography was performed on 30 patients. The most common abnormalities included thickened, irregular pleural line consistent with an inflammatory process, bilateral alveolar-interstitial syndrome (B-lines) and pulmonary consolidation (Table 3). Consolidation on lung ultrasonography was more likely to be identified among patients receiving mechanical ventilation than among those not receiving mechanical ventilation (75% v. 33%, odds ratio 6.0, 95% confidence interval 1.17–30.73).
Neurologic complications
Table 4 summarizes the short-term neurologic complications among 26 (10 non-ICU, 16 ICU) patients who underwent head CT during hospitalization. Three of 26 patients had an ischemic stroke, and 6 of 26 patients had intracerebral hemorrhage. The 3 patients who had an ischemic stroke did not have a history of previous ischemic stroke.
Extrapulmonary complications among patients hospitalized with COVID-19
Thrombotic complications and therapies
Thrombotic complications and therapies are shown in Table 4. Of the 13 (4 non-ICU, 9 ICU) patients who underwent Doppler ultrasonography, 2 ICU patients had deep venous thrombosis. Twenty-six (12 non-ICU and 14 ICU) patients underwent CT pulmonary angiography. Of these, 6 had pulmonary embolism (3 of 68 non-ICU patients and 3 of 32 ICU patients). Eleven (11.0%) patients received therapeutic anticoagulation during their hospitalization: 3 empirically for suspected COVID-19-associated hypercoagulable state, 7 for confirmed venous thromboembolic disease and 1 for venovenous extracorporeal membrane oxygenation circuit.
Cardiac and hemodynamic complications and therapies
Among 32 ICU patients, 24 (75.0%) received vasopressor therapy for a median of 9 (IQR 3.75–11.75) days. Four patients received hydrocortisone for refractory shock (Table 4).
Twenty-nine patients underwent echocardiography during their hospital stay (including both point-of-care and diagnostic). Overall, 4 (13.8%) patients had new left ventricular (LV) dysfunction and 9 (31.0%) patients had new right ventricular (RV) dysfunction (defined as either new systolic failure or pulmonary hypertension).
Hepatic complications
Alanine aminotransferase (ALT) levels were used as a surrogate marker for hepatic dysfunction in patients with COVID-19. The median of the highest ALT level across all study participants during hospital admission was 31 (IQR 22 to 82) U/L.
Renal complications and therapies
In 27 (27.0%) patients (non-ICU: 11/68 [16.2%]; ICU: 16/32 [50.0%]), an acute kidney injury developed. Among ICU patients, 5 received continuous renal replacement therapy for a median of 4 (IQR 2.0–6.5) days (Table 4). At the time of hospital discharge, 3 of 66 patients (4.5%) had persistent renal injury.
Secondary infections
Fifteen of 100 (15.0%) patients had an initially negative SARS-CoV-2 real-time polymerase chain reaction test, which was subsequently positive on repeat testing. In 13 patients, concomitant respiratory infections developed: 12 (12.0%) patients had bacterial respiratory infection and 1 (1.0%) patient had non-Candida fungal respiratory infection. Eight (8.0%) patients had at least 1 positive blood culture consistent with non-contaminant bacteremia, and 1 (1.0%) patient had Clostridioides difficile infection (Table 4).
Characteristics of patients transferred to ICU after hospital admission
Twelve of 100 patients (12.0%) were initially admitted to the ward but were subsequently transferred to ICU because of clinical decompensation shortly after hospitalization (median 0.5, IQR 0–2, d). Compared with patients who remained in a non-ICU location (n = 68), patients transferred to ICU were younger (median 63, IQR 53.75–68, yr v. median 76, IQR 60.3–84.8, yr; p = 0.01). Patients initially admitted to a non-ICU location who were later transferred to ICU were more likely to be febrile (≥ 38°C; 75.0% v. 28.4%; p = 0.003), have a respiratory rate of 24 breaths/min or greater (72.7% v. 34.3%; p = 0.02), and have an oxygen saturation level less than 92% (50.0% v. 11.9%; p = 0.02) on triage vital signs at presentation to hospital.
Patient outcomes
Patient outcomes and postdischarge imaging results to Dec. 31, 2020, are shown in Table 5. Thirty-four of 100 patients (34.0%) died in hospital, including 20 of 68 non-ICU patients (29.4%) and 14 of 32 ICU patients (43.8%). Persistent abnormalities were observed in 11 of the 19 patients who had CT of the thorax after discharge from hospital (median duration of follow-up 108, IQR 41.75–187.75, d). Similarly, 15 patients had repeat echocardiography after discharge from hospital (duration of follow-up 81, IQR 57–181, d), of which 1 had persistent LV dysfunction and 5 had persistent RV dysfunction.
Outcomes of patients hospitalized with COVID-19 after hospital discharge
Comparing survivors and nonsurvivors
We compared baseline characteristics of patients who did and did not survive to hospital discharge. Nonsurvivors were older (median 80, IQR 71.5–87.5, yr v. 68.5, IQR 54.0–77.3, yr; p < 0.01), more frequently presented with tachycardia (88.9% v. 24.1%; p < 0.01) and more frequently received supplemental oxygen on presentation to the emergency department (50.0% v. 26.6%; p = 0.02). On hospital admission, nonsurvivors were more frequently acidemic (pH < 7.35; 34.5% v. 17.8%; p = 0.04) and had a higher white blood cell count (10.6, IQR 7.1–14.3, ×109/L v. 6.7, IQR 4.8–8.7, ×109/L; p < 0.01), a lower hemoglobin level (124, IQR 113–135, g/L v. 134, IQR 123.3–145.8, g/L; p = 0.03) and a higher troponin level (28, IQR 14.5–75.5, ng/L v. 18, IQR 8.5–29.5, ng/L; p < 0.01).
Interpretation
In this prospective case series involving patients hospitalized with COVID-19, we found a range of pulmonary and extrapulmonary complications in both ICU and non-ICU patients. They had a high prevalence of neurologic complications, thrombotic complications, RV dysfunction and persistent cardiopulmonary pathology after hospital discharge.
Although COVID-19 was initially believed to precipitate isolated respiratory illness, evidence now characterizes it as a multisystem disease.3 The full spectrum of organ involvement and the associated outcomes in patients with COVID-19 remain relatively understudied.
In our study, nearly 10% of patients had a neurologic complication, including ischemic stroke and intracranial hemorrhage. Other studies reported the prevalence of ischemic stroke to be 2%–6%.11,12 One possible explanation for this phenomenon is that viral infections can result in an inflammatory cascade and endothelial injury that increase the risk of arterial thrombotic events.13,14 We also identified intracerebral hemorrhage as a potential sequela of COVID-19, with this complication developing in 6% of ICU patients in our cohort.15 Intracerebral hemorrhage is hypothesized to be due to the binding of SARS-CoV-2 to angiotensin-converting enzyme 2 receptors on endothelial cells of intracranial blood vessels, resulting in inflammation and disruption of vasculature integrity.16–18
In our cohort, thrombotic events (ischemic stroke, deep venous thrombosis and pulmonary embolism) occurred in 6% of non-ICU patients and 19% of ICU patients. Estimates of thrombotic events in hospitalized patients with COVID-19 have ranged from 5%19 to as high as 33%.20,21 Several mechanisms, including the complement pathway, neutrophil extracellular traps, inflammatory cytokines and endothelial dysfunction may explain why patients with COVID-19 could be hypercoagulable.22,23 Given the prevalence of thrombotic events, there may be value in routine surveillance as part of clinical care for patients with COVID-19. Although there is currently no evidence to support the routine use of anticoagulation, 24 antiplatelet therapy or systemic therapeutic anticoagulation may hold promise as a treatment for very specific subgroups of patients with COVID-19.25
There is a relative paucity of data describing the incidence of RV dysfunction in patients with COVID-19, which manifests as pulmonary hypertension, systolic failure or dilatation in these patients. In one observational study, the prevalence of pulmonary hypertension was 12% in patients with COVID-19 hospitalized in a non-ICU setting.26 In our study, we found that 31% of hospitalized patients had evidence of pulmonary hypertension. Although RV dysfunction has been described in patients with acute respiratory distress syndrome (ARDS), the prevalence of RV dysfunction in our study is higher than what has been reported in non-COVID-19 patients with ARDS.27–29 It is possible that RV dysfunction associated with COVID-19 may be pathologically distinct for several reasons. First, macroand microvascular thrombosis from deranged coagulation pathways could induce RV dysfunction. Second, “permissive hypoxia” as a treatment strategy may increase the prevalence of RV dysfunction.30,31
Similarly, we found evidence for long-term pulmonary complications associated with COVID-19 beyond hospital discharge. Abnormalities such as bronchiectasis, fibrosis or scarring were found in 57% of patients who underwent CT of the thorax after hospital discharge for clinical reasons in our cohort. Although pulmonary fibrosis in patients recovering from COVID-19 has been reported in several small cohorts,32 our study reinforces the notion that respiratory dysfunction can be prevalent and persistent.
These findings highlight the need to elucidate the true prevalence and potential mechanisms of extrapulmonary complications associated with COVID-19, particularly neurologic, thrombotic and cardiac manifestations. Although, at present, societal guidelines do not recommend the routine use of full-dose anticoagulation for patients with COVID-19, emerging clinical trial data suggest that systemic therapeutic anticoagulation may hold promise in improving outcomes among high-risk patients with COVID-19.33 Future studies should work to identify the patient phenotype for which the benefits of anticoagulation will outweigh the risks. The prevalence of acute and long-term RV dysfunction highlights the need to balance the respiratory support from positive pressure ventilation with the adverse mechanical effects it frequently imposes on RV function. Lastly, whereas the acute inpatient management of COVID-19 has garnered attention among the scientific community, future research should prioritize characterization of the long-term pulmonary and extrapulmonary complications of COVID-19.
This study has several strengths. We included a cohort of ICU and non-ICU patients across a spectrum of disease severity. Standardized diagnostic microbiologic methods were used to define SARS-CoV-2 positivity. Consecutive prospective enrolment reduced selection bias and improved the fidelity of data collection. We also present granular data on the pulmonary and extrapulmonary manifestations of COVID-19 and secondary infections among patients with COVID-19 in a health care setting. We provide data on the clinical outcomes of patients after hospital discharge. Finally, this study also provides a detailed experience of patients hospitalized with COVID-19 starting from hospital admission to beyond hospital discharge.
Limitations
Limitations of any observational study such as this one include the inability to draw conclusions about causality of any specific exposure (e.g., therapy or management strategy) with outcomes. In this study, several diagnostic tests (e.g., ultrasound studies, CT scans, echocardiograms) were performed on a subgroup of patients who may have differed from those not assessed owing to testing and treatment by indication. Furthermore, our cohort reflects patients at 2 Ontario hospitals during the first wave of the COVID-19 pandemic. Larger, multicentre cohort studies are required to provide data that are generalizable beyond the location of this study and the first wave of the pandemic.
Conclusion
This study provides further evidence that COVID-19 is a multisystem disease that results in neurologic, cardiac and thrombotic complications in the acute phase, as well as pulmonary complications that persist beyond the hospitalization. These findings underscore the need to prioritize research on the long-term outcomes and management of COVID-19 survivors.
Footnotes
Competing interests: Ian Ball declares a research grant from the Lawson Health Research Institute Internal Research Fund. He is the critical care lead for Ontario West and sits on the Ontario Critical Care COVID-19 Command Table. Mike Nicholson received teaching honoraria unrelated to acute COVID-19 from AstraZeneca, Horizon Therapeutics and Vertex Pharmaceuticals, and has served on a cystic fibrosis drug efficacy advisory board for Horizon and as a moderator for cystic fibrosis gene modulator launch educational seminars for Vertex Pharmaceuticals. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Kimia Honarmand and John Basmaji conceived and designed the study, and oversaw all study conduct. Claudio Martin, Karen Bosma, Marat Slessarev, Ian Ball, Tina Mele and Sameer Elsayed contributed to the conception and design of the study. Ian Ball, Kimia Honarmand, John Basmaji and Karen Bosma obtained funding for the study. Kimia Honarmand, John Basmaji, Kyle Fiorini, Debarati Chakraborty, Daniel Gillett, Karishma Desai and Danielle LeBlanc participated in data collection and database management. Kimia Honarmand and John Basmaji conducted the data analysis. Claudio Martin, Karen Bosma, Marat Slessarev, Ian Ball, Sameer Elsayed, Alejandro Lazo-Langer, Mike Nicholson and Robert Arntfield contributed to the interpretation of the results. Kimia Honarmand, John Basmaji, Karen Bosma, Marat Slessarev and Claudio Martin prepared the initial draft of the manuscript. All authors contributed to manuscript preparation and revision, approved the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was funded by a grant provided by the Lawson Health Research Institute’s Internal Research Fund (IRF-0665). The funders had no role in the collection, analysis, interpretation or dissemination of the study data.
Data sharing: All data will be available on request.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/3/E675/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors