Abstract
Background: The prevalence of adult chronic kidney disease and cardiovascular comorbidities in Canadian Indigenous communities is largely unknown. We conducted a study to document the prevalence of chronic kidney disease and concurrent diabetes mellitus, hypertension and dyslipidemia in a First Nations population in northwest Ontario.
Methods: In this observational study, we used retrospective data collected from regional electronic medical records of 16 170 adults (age ≥ 18 yr) from 26 First Nations communities in northwest Ontario from May 2014 to May 2017. Demographic and laboratory data included age, gender, prescribed medications, estimated glomerular filtration rate, urine albumin:creatinine ratio, low-density lipoprotein cholesterol (LDL-C) level and glycated hemoglobin (HbA1c) concentration. We identified patients with diabetes by an HbA1c concentration of 6.5% or higher, or the use of a diabetic medication, those with dyslipidemia by an elevated LDL-C level (≥ 2.0 mmol/L) or use of lipid-lowering medication, and those with hypertension by use of antihypertensive medication.
Results: Of the 16 170 adults residing in the communities, 5224 unique patients (32.3%) had renal testing (albumin:creatinine ratio and/or estimated glomerular filtration rate). The age-adjusted prevalence of chronic kidney disease was 14.5%, and the prevalence of stage 3–5 chronic kidney disease (estimated glomerular filtration rate < 60 mL/min) was 7.0%. Most patients with chronic kidney disease (1487 [80.0%]) had at least 1 cardiovascular comorbidity. A total of 1332 patients (71.6%) had diabetes, 1313 (70.6%) had dyslipidemia, and 1098 (59.1%) had hypertension; all 3 comorbidities were present in 716 patients (38.5%).
Interpretation: We document a high prevalence of advanced chronic kidney disease in this First Nations population, 7.0%, double the rate in the general population. High rates of cardiovascular comorbidities were also common in these patients with chronic kidney disease, which places them at increased risk for cardiovascular disease.
Planning chronic disease management services for a population requires knowledge of the existing comorbid diseases to assist in both primary and secondary prevention. For rural and remote populations spread over a vast region who must come to a central health hub for services such as dialysis initiation, it is essential. Information on the prevalence of chronic kidney disease, a precursor to end-stage renal disease, and its risk factors for progression is a necessity given the impact on quality of life and costs involved.1–3 In response to rising rates of end-stage renal disease and chronic kidney disease, First Nations community chiefs in northwest Ontario requested a better understanding of the burden of chronic kidney disease and related comorbidities.
Indigenous Canadians experience a high burden of chronic kidney disease.4–6 A 2001 screening study in Canadian First Nations communities suggested an estimate in adults of up to 30%.6 First Nations populations in Canada are more likely than non-Indigenous populations to progress to end-stage renal disease, and their rate of death related to chronic kidney disease is 77% higher than that in the non-Indigenous population.7–10 The prevalence of diabetes mellitus, a major risk for progressive chronic kidney disease, is higher in Indigenous population than in non-Indigenous populations, and it occurs at a younger age and is associated with increased mortality, cardiovascular disease and lower limb amputation.11,12 Uncontrolled hypertension is also major risk factor for progression to end-stage renal disease and, together with diabetes, dyslipidemia and chronic kidney disease, contributes to a high rate and severity of cardiovascular disease, the leading cause of death in this population. 13,14 The purpose of this study was to document the prevalence of chronic kidney disease and concurrent diabetes, hypertension and dyslipidemia in a First Nations population in northwest Ontario.
Methods
Setting
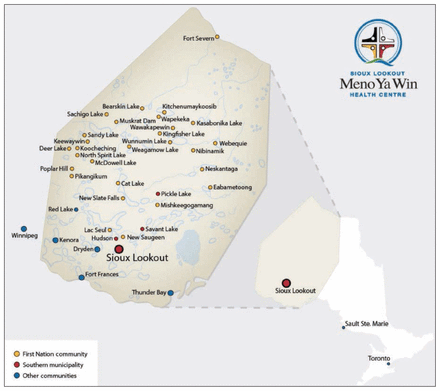
The setting was northwest Ontario, where 26 remote First Nations communities are supported in primary care by the Sioux Lookout First Nations Health Authority, which oversaw the study (Figure 1).
Sioux Lookout First Nations Health Authority and neighbouring communities in northwestern Ontario. Reproduced with permission of the Sioux Lookout Meno Ya Win Health Centre.
Design
For this descriptive study, we used retrospective clinical data from a regional electronic medical record system from May 2014 to May 2017, during which most patients with a chronic condition had a primary care encounter. Most patients with chronic conditions have at least 1 primary care visit in 2 years.15,16 We chose a 3-year period to capture a robust number of patients with relevant prescriptions or investigations to estimate both incident and prevalent cases. Thirty community physicians have used the OSCAR (Open Source Clinical Application and Resource) electronic medical record system exclusively since 2013. The OSCAR database includes all community members accessing primary care in their community or in Sioux Lookout. All primary care visits, medications and investigations are recorded in the electronic medical record.17 The study population comprised all patients aged 18 years or more who had at least 1 clinical encounter between May 2014 and May 2017 and received a prescription or investigation for renal function, diabetes, hypertension or dyslipidemia.
Sources of data
Data were extracted by the Sioux Lookout First Nations Health Authority information technology department (J.H.) and verified by its public health service epidemiologist (C.M.). Data services are overseen by an information management committee of the Sioux Lookout Regional Physicians Services. Data collection followed Canadian Primary Care Sentinel Surveillance Network methods and validated case definition; the network also provided online support for local data extraction.18,19
Clinical data in the OSCAR electronic medical record are input by individual health care providers at the time of patient visits. Laboratory data are automatically updated by the Sioux Lookout Meno Ya Win Health Centre laboratory, which preforms all inpatient and outpatient testing. Medication data in the electronic medical record are regularly updated by the 30 treating physicians and are linked to regional pharmacies. Outpatient prescribing is done exclusively through the electronic medical record by regional primary care physicians. The extracted data accurately reproduced patient chart information in 20 randomly selected charts, several from either data subset (diabetes medications, low-density lipoprotein cholesterol [LDL-C] values). We used Anatomic Therapeutic Classification codes20 to select all disease-related medication data (antihypertensive medications [C02], peripheral vasodilators [C04], β-blocking agents [C07], calcium-channel blockers [C08], agents acting on the renin–angiotensin system [C09], lipid-modifying agents [C10A-B], insulin and analogues [A10A] and blood-glucose-lowering drugs [A10B]).
Diagnostic codes were not available as they are not automatically recorded in the electronic medical record. Laboratory investigations and medication prescribing were believed to be complete; they can be performed only through the electronic medical record system and were thought to be a reliable reflection of disease prevalence.18
Demographic and laboratory data included age, gender, medications, estimated glomerular filtration rate, albumin: creatinine ratio, LDL-C level and glycated hemoglobin (HbA1c). The most recent laboratory value was used. We defined chronic kidney disease according to International Society of Nephrology criteria: estimated glomerular filtration rate less than 60 mL/min per 1.73 m2 or urine albumin: creatinine ratio 3 mg/mmol or greater.21 We identified patients with hypertension through prescriptions for antihypertensive medications. Patients with diabetes were identified by an HbA1c concentration of 6.5% or less, or the use of a diabetic medication. We identified patients with dyslipidemia by elevated LDL-C level (≥ 2.0 mmol/L) or the use of a lipid-lowering medication.12
Statistical analysis
We calculated the prevalence of chronic kidney disease in patients with at least 1 albumin:creatinine ratio or estimated glomerular filtration rate investigation during the study period, age adjusted to the 2016 Canadian census population. We calculated the comorbid prevalence of diabetes, hypertension and dyslipidemia using the population of patients with chronic kidney disease as the denominator. We calculated the presence of multiple comorbidities as the proportion of patients with chronic kidney disease affected. Data are presented as mean and standard deviation for continuous variables and proportions for discreet variables.
Ethics approval
The research was supervised and the data were maintained by the Sioux Lookout First Nations Health Authority, which represents communities in health programming and surveillance in accordance with the principles of ownership, control, access and possession (OCAP).22 Ethics approval was granted by the Sioux Lookout Meno Ya Win Research Review and Ethics Committee and the Lakehead University Research Ethics Board.
Results
The total population of northwest Ontario in 2017 was 24 789, including 16 170 adults residing in the 26 communities.17 Renal testing (estimated glomerular filtration rate or albumin: creatinine ratio) had been performed in 5224 unique patients (32.3%) from May 2014 to May 2017. Of the 16 170 adults, 4578 (28.3%) had at least 1 estimated glomerular filtration rate measurement, and 2462 (15.2%) at least 1 albumin:creatinine ratio measurement, with overlap between these 2 groups. Abnormal results occurred in 806 (17.6%) of the estimated glomerular filtration rate tests and 1433 (58.2%) of the albumin:creatinine ratio tests.
We identified 1859 patients with chronic kidney disease. The resulting age-adjusted chronic kidney disease prevalence was 14.7%. The mean age of these patients was 55 years, 14 years older than the average adult in the electronic medical record (Table 1). Of the 5224 adults who had renal testing, 2945 (56.4%) were women, and they had a burden of chronic kidney disease similar to that of men. The age-adjusted prevalence of reduced renal function (stage 3–5 chronic kidney disease, estimated glomerular filtration rate < 60 mL/min) was 7.0%, and that of albuminuria with normal renal function (stage 1–2 chronic kidney disease) was 7.7% (Table 2).
Sex and age of adult population, patients with renal testing and patients with chronic kidney disease in 26 First Nations communities in northwest Ontario
Prevalence of chronic kidney disease
A total of 1487 patients with chronic kidney disease (80.0%) had at least 1 cardiovascular comorbidity (diabetes, hypertension or dyslipidemia), 1078 (58.0%) had 2 cardiovascular comorbidities, and 716 (38.5%) had all 3 cardiovascular comorbidities (Table 3). Diabetes was most often coprevalent (1332 [71.6%]), followed by dyslipidemia (1313 [70.6%]) and hypertension (1098 [59.1%]).
Laboratory values, drug use and comorbidities of patients with chronic kidney disease
Almost one-third (539 [29.0%]) of patients with chronic kidney disease were not screened for lipid levels during the study period, 980 (52.7%) were receiving a statin, and 688 (37.0%) met the recommended LDL-C goal.
Interpretation
Renal screening occurred in 32% of the adult population of 26 First Nations communities of northwest Ontario during the 3-year study period; 15% had albumin:creatinine ratio testing, and 28% had estimated glomerular filtration rate testing. There was a high coprevalence of diabetes (72%), dyslipidemia (71%) and hypertension (59%). A substantial proportion of patients with chronic kidney disease, 38%, had all 3 cardiovascular comorbidities. The age-adjusted prevalence of chronic kidney disease was 14.7%; half of these patients had stage 3–5 disease.
The prevalence of chronic kidney disease in our population is slightly higher than the 2013 estimated Canadian rate of 12.5%23 but lower than that in recent population screening reports among Manitoba First Nations and southern Ontario First Nations communities of 25% and 30%, respectively.4–7,23 Our prevalence of stage 3–5 chronic kidney disease is higher, at 7% versus 3%–5%.5,6,23 These variations are likely due to methodological differences, primarily the use of a screened population versus a practice-based cohort. Studies using data arising from patient care cohorts showed a higher prevalence of stage 3–5 disease (7%–8%).14,24 Our methodology approximated that of a 2019 study of the prevalence of advanced chronic kidney disease in the general Canadian population, with electronic medical record data extraction from primary care practices.24 Those authors used only estimated glomerular filtration rate results (no albumin:creatinine ratio), with findings equivalent to ours (prevalence of stage 3–5 disease of 7%). Similarly, in a study using data from health administrative databases from 1980–2005 (before the advent of the electronic medical record) in Saskatchewan, the prevalence of advanced disease was 8%.14
The high coprevalence of chronic kidney disease and diabetes in our population, 72%, is not surprising given the high burden of diabetes in First Nations populations. This rate is higher than that reported in a recent Manitoba screening study, 60%.5,25,26 The coprevalence of dyslipidemia, 71%, is higher than estimates of 30% in the overall adult First Nations and Canadian populations.27,28 The average LDL-C level in our patients with chronic kidney disease was 2.1 mmol/L, lower than the value of 2.4 mmol/L observed in the 2012 Canadian First Nations Diabetes Clinical Management and Epidemiologic (CIRCLE) study among 885 First Nations people with diabetes.4 The coprevalence of hypertension in our population, 59%, is higher than the rates of 27% identified in the 2016 Manitoba study5 and 29.5% in a 2011 study of 555 First Nations community members in southern Ontario.6 Only 56% of our patients with chronic kidney disease who had hypertension were receiving treatment with angiotensin-converting-enzyme inhibitors or angiotensin receptor blockers. The prevalence of these comorbidities with chronic kidney disease confers additional cardiovascular risk.13,14 The CIRCLE study investigators noted that progression from albuminuria to advanced chronic kidney disease was associated with duration of diabetes and coprevalence of hypertension and dyslipidemia in First Nations patients.4
Chronic kidney disease independently increases the risk of adverse cardiovascular events.29,30 The high coprevalence of other cardiovascular comorbidities in this First Nations population signals the need for broad assessment and risk management. 14,30 These findings will play an important role in planning dialysis services and in secondary prevention of progression of chronic kidney disease and cardiovascular disease.
Limitations
Our study has limitations owing to the real-world nature of the data, with relevant testing of only 32% of the adult population. Our noted prevalence of chronic kidney disease is likely an underestimate when compared to screening studies that include universal albumin:creatinine ratio and estimated glomerular filtration rate testing.5,6 Urine albumin:creatinine ratio testing gives the highest yield of chronic kidney disease detection but is not universally used in general practice; it was ordered in only 15% of our population. Urine screening at point-of care-testing (dipstick) is common but is not systematically recorded in this electronic medical record (available in clinical notes only). Our study is therefore limited in identifying early-stage chronic kidney disease, but the high prevalence of advanced chronic kidney disease in this population is notable, particularly as only 28% of patients received estimated glomerular filtration rate testing. Retrospective data collection based on electronic medical record is limited to available and extractable data. Information on blood pressure measurements, body mass index, and rates of smoking and cardiovascular disease were not available, nor were diagnosis or physician billing data.
Conclusion
We document a substantial prevalence of stage 3–5 chronic kidney disease and associated cardiovascular comorbidities in this First Nations population. Coprevalence of these major chronic diseases in this population increases cardiovascular risk, and robust clinical assessment is warranted. These findings will help health care providers identify people at high risk for cardiovascular and progressive kidney disease.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Sheldon Tobe initiated the study. Len Kelly designed the study. Janet Gordon and Sheldon Tobe oversaw the study, and Janet Gordon oversaw the study process, including community permission and data extraction. Cai-lei Matsumoto supervised the data collection and analyzed the data. Christopher Olivier and Josh Hopko collected the data. Hannah Willms and Christopher Olivier performed the literature searches. Len Kelly, Sheldon Tobe and Cai-lei Matsumoto interpreted the data. Len Kelly, Yoko Schreiber, Sharen Madden and Sheldon Tobe drafted the manuscript, and Yoko Schreiber and Sharen Madden critically revised it for important intellectual content. All of the authors approved the final version to be published and agreed to act as guarantors of the work.
Funding: This work was supported by the Northern Ontario Academic Medicine Association.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/7/3/E568/suppl/DC1.
References
- Copyright 2019, Joule Inc. or its licensors