Abstract
Background The Goals of Care Designation (GCD) is a medical order used to communicate the focus of a patient’s care in Alberta, Canada. In this study, we aimed to determine the association between GCD type (resuscitative, medical or comfort) and resource use during hospitalization.
Methods This was a prospective cohort study of newly hospitalized inpatients in Alberta conducted from January to September 2017. Participants were aged 55 years or older with chronic obstructive pulmonary disease, congestive heart failure, cirrhosis, cancer or renal failure; aged 55–79 years and their provider answered “no” to the “surprise question” (i.e., provider would not be surprised if the patient died in the next 6 months); or aged 80 years or older with any acute condition. The exposure of interest was GCD. The primary outcome was health care resource use during admission, measured by length of stay (LOS), intensive care unit hours, Resource Intensity Weights (RIWs), flagged interventions and palliative care referral. The secondary outcome was 30-day readmission. Adjusted regression analyses were performed (adjusted for age, sex, race and ethnicity, Clinical Frailty Scale score, comorbidities and city).
Results We included 475 study participants. The median age was 83 (interquartile range 77–87) years, and 93.7% had a GCD at enrolment. Relative to patients with the resuscitative GCD type, patients with the medical GCD type had a longer LOS (1.42 times, 95% confidence interval [CI] 1.10–1.83) and a higher RIW (adjusted ratio 1.14, 95% CI 1.02–1.28). Patients with the comfort and medical GCD types had more palliative care referral (comfort GCD adjusted relative risk (RR) 9.32, 95% CI 4.32–20.08; medical GCD adjusted RR 3.58, 95% CI 1.75–7.33) but not flagged intervention use (comfort GCD adjusted RR 1.06, 95% CI 0.49–2.28; medical GCD adjusted RR 0.98, 95% CI 0.48–2.02) or 30-day readmission (comfort GCD adjusted RR 1.00, 95% CI 0.85–1.19; medical GCD adjusted RR 1.05, 95% CI 0.97–1.20).
Interpretation Goals of Care Designation type early during admission was associated with LOS, RIW and palliative care referral. This suggests an alignment between health resource use and the focus of care communicated by each GCD.
Health care is moving away from using binary “resuscitate” and “do not resuscitate” medical orders, to more nuanced ways of communicating patients’ treatment preferences.1 Order frameworks help communicate patients’ care goals to health care providers.2,3 Alberta’s Goals of Care Designation (GCD) order, introduced in 2014, was Canada’s first province-wide order framework.3,4 These orders communicate the general focus of a person’s care, specific clinically indicated health interventions, transfer decisions and locations of care that might be used in service of a person’s goals. Alberta’s GCD order describes 3 general approaches to care, with subcategories to communicate nuances within: resuscitative GCD, medical care GCD and comfort care–focused GCD.5 A comfort care GCD communicates a focus on comfort care that provides relief from symptoms, without cure or control of the underlying condition.5 When possible, this comfort approach is applied in the person’s home, residence or hospice without transfer to hospital unless symptom relief cannot be achieved in that setting. Medical care GCD communicates a focus on cure or control of the underlying conditions, without the use of resuscitative interventions (e.g., intensive care unit [ICU] admission, cardiopulmonary resuscitation or intubation). Resuscitative GCD communicates a focus on cure and control of the patient’s condition, including the use of resuscitative interventions if required.5 A GCD is established after consultation between the most responsible health care provider and patient or their alternate decision-maker. A paper copy of a patient’s GCD is kept in a plastic “Green Sleeve,” which is recognized across the care continuum, including by emergency medical services, and used to transport the information between care settings. During transfers, the most responsible health care provider is responsible for reviewing the GCD with the patient and entering the current desired approach to care into the health record in use at that location. Goals of Care Designations are fluid, changing with the clinical situation and patient goals. Patients and providers are encouraged to communicate with each interaction to ensure that the GCD that best reflects the patient’s wishes in their current clinical context is documented.
We were interested in determining whether care provided is concordant with the GCD recorded and the cost implications of GCDs. Patients who have chosen a comfort GCD might be expected to have lower rates of invasive care, make greater use of palliative care and have lower associated costs.6,7 This has been the finding of some studies performed in Ontario, Canada, and several states in America,8–11 but US-based studies have also found no association between such orders and hospitalization, ICU admission, inpatient costs or place of death.9,12 Patient population and location may influence these associations.9,10,12 In this prospective cohort study, we sought evidence that patients’ GCD type (indicting preference for care) is reflected in the care provided. To do this, we examined the association between GCD type early in seriously ill patients’ new hospitalization and health care resource use during that admission. We hypothesized that after adjusting for illness severity, inpatients with a resuscitative GCD would have higher resource use than patients with a comfort GCD (appropriately so, as providers should use all possible medical intervention for patients with a resuscitative GCD, but not for patients with a comfort GCD). As a secondary outcome, we examined the association between GCD type at discharge and 30-day readmission. As a comfort GCD indicates a patient’s preference to avoid transfer, we hypothesized that patients with a comfort GCD discharged home would be less likely to be readmitted than patients with a resuscitative GCD discharged home.
Methods
This was a multicentre, prospective cohort study involving adults admitted to acute care in Alberta, Canada. The province-wide cohort was collected as a new audit cycle of the prior Canada-wide Audit of Communication, Care Planning, and Documentation (ACCEPT) study.13,14 Briefly, the ACCEPT study examined advance care planning activities of patients and quantified the agreement between expressed preference for care and documented orders. This study examines the association between documented GCD and subsequent health care resource use in hospital and 30 days postdischarge. By necessity, the current study followed the patient inclusion criteria and recruitment procedures laid out by the ACCEPT study.13,14 STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) was used as the reporting guideline. Additional study protocol details are provided in Appendix 1, available at www.cmajopen.ca/content/10/4/E945/suppl/DC1.
Setting
Consecutive, consenting participants were recruited on weekdays from 36 inpatient units at 8 hospitals in Alberta (Calgary area [3 hospitals, 13 units], Edmonton area [3 hospitals, 21 units] and Lethbridge area [2 hospitals, 2 units]; 2017 populations of 1.3 million, 1 million and 100 000, respectively). Sites were purposively chosen to include tertiary teaching and community hospitals (specialized and general units) that typically serve adults admitted acutely with an advanced chronic illness (i.e., sites were enriched for patients likely to meet the inclusion criteria; see “Participants”). Recruitment ran from January to September 2017, the time needed to accrue 200 participants in each of Calgary and Edmonton, and 100 in Lethbridge. The sample size was chosen to yield adequate power for the Alberta ACCEPT audit and its primary outcome (patient awareness of their GCD). From the perspective of this study’s outcomes, the cohort collected is a convenience sample.
Participants
Eligible patients were admitted to hospital from the community and were aged 55 years or older with a diagnosis of advanced chronic obstructive pulmonary disease, congestive heart failure, cirrhosis, cancer or renal failure; aged 55–79 years and their provider answered “no” to the “surprise question”15,16 (i.e., provider would not be surprised if the patient died in the next 6 months); or aged 80 years or older and admitted because of any acute condition (see Appendix 1 for all clinical criteria used). Prior studies showed that applying these criteria produces a cohort with an aggregate 6-month mortality rate of ~50%.17,18 This study, and the ACCEPT study,13 focused on this patient population because they are a group for whom GCD documentation is highly relevant and should have occurred (or be occurring). Patients were ineligible during the first 48 hours of admission to allow time for symptoms to have abated enough for patients to participate in the survey, and after 120 hours because the ACCEPT study focused on communication during early hospitalization.13,14 Research nurses screened the charts of all patients admitted to the participating hospital units to identify potentially eligible patients and spoke with charge and bedside nurses to evaluate exclusion criteria. Patients were excluded if they were unable to communicate in English, were cognitively impaired or had other communication difficulties (e.g., auditory, visual or dysphasia) that could not be accommodated sufficiently to allow for them to consent and complete the study questionnaire. Patients were excluded if they were newly diagnosed with a life-limiting illness during the admission or imminently dying (i.e., a prognosis of death is expected to occur within hours to days).19,20 Exclusion criteria were not evaluated using formal assessment tools but based on the judgment of a bedside nurse and/or attending physician (for additional description of the screening and recruitment procedures followed, see Appendix 1).
Per requirements of our ethics board, the bedside nurse approached eligible patients to ask if they would meet with the research nurse. If the patient agreed, the research nurse would invite the patient to participate in the study and obtained consent. Consenting patients were administered the study questionnaire in person by the research nurse. On the same day, enrolment data were abstracted from patients’ medical charts (electronic and physical) by the research nurse.
Source of data
The participant questionnaire administered by the research nurse was the data source for self-reported race and ethnicity, education, residence and Clinical Frailty Scale score.21 Medical chart (electronic and physical) abstraction performed by the research nurse at enrolment was the data source for GCD before admission, GCD at study enrolment, patient age and sex. Information on GCDs can be found in several areas of a patient’s medical record (paper and electronic), and a patient can have multiple GCDs over time (even on the same day). To address this, the research nurses were carefully trained to inspect all possible locations for GCD order forms and to record all GCD orders found along with their date and time, and location. The GCD with the latest date and time (most recent relative to when enrolment occurred) was taken to be the GCD at study enrolment. Medical chart abstraction (electronic and physical) was repeated by the research nurse 3 months later to determine the GCD at discharge from the index hospital admission (during which the patient was enrolled), specialist palliative care referral during admission, and index admission discharge outcome and location. Patients can change GCD type during hospitalization; therefore, we determined the percent of the index admission duration for which the GCD type at enrolment (i.e., the predictor in the analysis of the primary outcomes) was applicable. Medical chart abstraction followed a guideline provided to the research nurses. The Canadian Institute for Health Information’s (CIHI’s) Discharge Abstract Database (DAD) was the data source for length of stay (LOS), ICU admission, CIHI Resource Intensity Weights (RIWs) and CIHI flagged intervention use, and was used to assess comorbidities using International Classification of Disease, Tenth Revision (ICD-10) diagnosis codes (codes 1–25). Data from DAD were obtained for the index hospital admission and all readmissions to any Alberta hospital occurring within 6 months of study enrolment, allowing for evaluation of 30-day readmission. DAD ICD-10 codes for the index admission were used to determine patients’ weighted Charlson Comorbidity Index score to adjust for comorbidities.22,23 For patients still in hospital at 3 months, discharge outcome and location were replaced using DAD-sourced data if there was a change between 3 and 6 months from study enrolment (affecting 15 participants).
Outcomes
The primary outcome was health care resource use during admission, measured by LOS, ICU admission, CIHI RIW, CIHI flagged interventions and specialist palliative care referral. The secondary outcome was 30-day readmission. Resource Intensity Weights express an inpatient’s expected hospital resource use relative to the average typical inpatient case. They are assigned based on the CIHI case-mix group to which an individual belongs as well as their age, health status and discharge status.24–26 By definition, the average typical inpatient case is assigned an RIW of 1.0. To provide context, RIW was expressed in terms of relative cost by multiplying RIW by Alberta’s Cost of a Standard Hospital Stay for 2017–2018 ($7893).27 The Canadian Institute for Health Information’s list of 16 flagged interventions (identified by 433 procedure codes) indicates potentially complex, resource-intensive and high-cost inpatient interventions, many of which are used in the process of providing life support.28,29 Specialist palliative care is an indicator of high-quality care for patients with advanced illness30 and is associated with lower hospital care costs.31,32 The exposure of interest was chart-recorded GCD at study enrolment and chart recorded GCD at discharge (for 30-day readmission).
Statistical analysis
Categorical variables were evaluated for association with GCD type using the Fisher exact test; continuous variables were evaluated using the Kruskal–Wallis test. To compare primary and secondary outcomes by GCD type, we used unadjusted and adjusted regression models as appropriate based on outcome parameterization and distribution. For dichotomous outcomes (palliative care referral [yes or no], flagged intervention use [any or none] and 30-day readmission [yes or no]), we used modified Poisson regression33 with robust (sandwich) estimation of variance. For LOS and RIW, we used truncated negative binomial regression and γ (log-link) regression, respectively. Goals of Care Designation type at enrolment (for primary outcomes) or discharge (for 30-day readmission) was modelled as a nominal exposure, with resuscitative GCD as the reference. All adjusted models were adjusted for age, sex, race and ethnicity (white, other), Clinical Frailty Scale score (mildly frail, severely frail), weighted Charlson Comorbidity Index score (low, moderate–high) and city. The model for 30-day readmission was additionally adjusted for discharge location. Discharge location is expected to be an effect modifier in the relation between GCD type and 30-day readmission; therefore, we performed a stratified analysis on just those patients discharged to home (the only discharge location with sufficient observations to warrant this analysis). Variables were chosen a priori based on previous work.13 Analyses were performed in R version 4.0.0.34 All tests were 2-sided, and p < 0.05 defined significance.
Ethics approval
The health research ethics boards of the University of Calgary and the University of Alberta approved this study.
Results
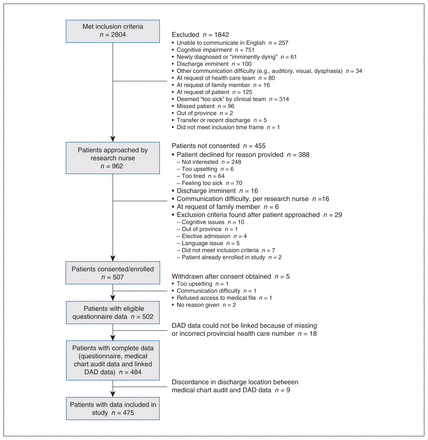
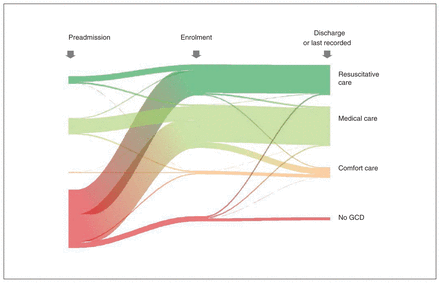
We included 475 study participants (Figure 1). The median age was 83 (interquartile range [IQR] 77–87) years, 226 (47.6%) were male and 439 (92.4%) self-reported as “white” (Table 1). Most participants, 294 (61.9%), had relatively few comorbidities (weighted Charlson Comorbidity Index score 0–2); only 25 (5.3%) were “severely frail” (Clinical Frailty Scale score 7–9). Most participants (n = 332, 69.9%) did not have a GCD in their medical record before admission (Figure 2; Appendix 1, Supplementary Table 1). By the time of study enrolment 48–120 hours into the admission, 445 (93.7%) participants had a GCD. Medical GCDs were most common (n = 248, 52.2%), followed by resuscitative GCDs (n = 177, 37.3%), no GCD (n = 30, 6.3%) and comfort GCDs (n = 20, 4.2%). A total of 20.0% (n = 4/20) of participants with a comfort GCD at enrolment died during the index admission, and 9.3% and 3.4% of participants with a medical and resuscitative GCD, respectively, died during admission (Table 1). The mean percentage of time the GCD type at enrolment reflected the GCD type at any point during the index admission was 98% (standard deviation [SD] 8%) for resuscitative GCD, 97% (SD 9%) for medical GCD, 88% (SD 15%) for comfort GCD and 96% (SD 10%) for no GCD.
Flow chart showing recruitment of participants. Note: DAD = Discharge Abstract Database.
Demographic and medical characteristics of study participants at enrolment
Ribbon plot showing study participants’ change in Goals of Care Designation (GCD) type over time, from pre–hospital admission to study enrolment, to last recorded at discharge, death or end of study. Colour corresponds to GCD type (resuscitative care GCD: dark green; medical care GCD: light green; comfort care GCD: orange; no GCD: red). Line thickness corresponds to the proportion of the sample.
Health resource use during admission
Patients with a resuscitative GCD at enrolment had a median LOS of 7 (IQR 5–12) days and RIW of 1.0 (IQR 0.7–1.6). Nine of 177 (5.1%) experienced palliative care referral, and 38 of 177 (21.5%) had a flagged intervention (Table 2). Patients with a comfort GCD had a median LOS of 10 (IQR 7–16) days and RIW of 1.5 (IQR 1.0–1.9). Eighteen of 20 (90.0%) experienced palliative care referral, and 7 of 20 (35.0%) had a flagged intervention (the latter were primarily symptom-relieving interventions, not life-supporting). Four patients were admitted to the ICU after enrolment (none of comfort or medical GCD). Using RIW and Alberta’s cost of a standard hospital stay for 2017–2018, the average estimated cost of hospitalization was $7893.00 for patients with a resuscitative GCD and $11 839.50 for patients with a comfort GCD. With respect to health resource use outcomes, patients with a medical GCD were intermediate to those with a resuscitative or comfort GCD; patients with no GCD were most like those with a resuscitative GCD.
Health care resource use during hospitalization, by GCD type
Adjusted regression models examining the association between GCD type and LOS and RIW showed that only patients with a medical GCD were significantly different from those with a resuscitative GCD (Table 3). Specifically, patients with a medical GCD had an LOS 1.42 times that of those with a resuscitative GCD (95% CI 1.10–1.83), and they had an RIW 14% higher (adjusted ratio 1.14, 95% CI 1.02–1.28). Patients with a comfort GCD, although not significantly different, had similar effect size estimates (LOS 1.26 times, 95% CI 0.68–2.36; RIW 13% higher, 95% CI 0.87–1.48). In the adjusted model for inpatient palliative care referral, patients with a comfort GCD and patients with a medical GCD were more likely to be referred than patients with a resuscitative GCD (comfort GCD adjusted relative risk [RR] 9.32, 95% CI 4.32–20.08, medical GCD adjusted RR 3.58, 95% CI 1.75–7.33).
Association between GCD at enrolment and health care resource use during hospitalization
Discharge outcome and hospital readmission 30 days after discharge
Most participants (n = 437/475, 92.0%) were discharged from the index admission during the study’s 6-month follow-up period (Appendix 1, Supplementary Table 2). Thirty-five participants (7.4%) died during the admission; 3 (0.6%) were still in acute care at 6 months. Discharged participants were discharged to home (n = 309, 70.7%), retirement residence (n = 63, 14.4%), long-term care facility (n = 32, 7.3%), hospice (n = 17, 3.9%), rehabilitation facility (n = 14, 3.2%) or an unspecified location (n = 2, 0.5%) (Appendix 1, Supplementary Table 2). Participants’ last recorded GCD (at discharge, death or end of study) was associated with discharge outcome and location (Fisher exact test, p < 0.001) (Appendix 1, Supplementary Table 2). Of participants who died in hospital, 26 of 35 (74.3%) had a comfort GCD at the time of death. All 17 participants discharged to hospice had a comfort GCD, which is typically required for hospice admission in Alberta. Most participants (86.5%) did not change their GCD type between study enrolment and discharge (Figure 2). Of the 64 that did, 34 (53.1%) changed from a medical to comfort GCD, and 9 (14.1%) changed from a resuscitative to medical GCD.
Among the participants discharged from acute care and followed for 30 days or longer, 76 of 437 (17.4%) were readmitted within 30 days. By GCD type, 26 of 168 (15.5%) patients with a resuscitative GCD were readmitted, 47 of 219 (21.5%) with a medical GCD, 2 of 33 (6.1%) with a comfort GCD and 1 of 17 (5.9%) with no GCD (Table 4). In regression models examining the association between GCD type at discharge and hospital readmission in 30 days, patients with a medical or comfort GCD were not significantly different from patients with a resuscitative GCD (medical GCD adjusted RR 1.05, 95% CI 0.97–1.20; comfort GCD adjusted RR 1.00, 95% CI 0.85–1.19). Because discharge location can cause a patient to be more or less likely to be readmitted to hospital, we performed 1 stratified analysis on those 309 patients discharged home. Again, discharge GCD was not associated with 30-day readmission (Table 4; medical GCD adjusted RR 1.06, 95% CI 0.98–1.16; comfort GCD adjusted RR 1.01, 95% CI 0.74–1.36). However, only 5 of 33 (15.2%) patients with a comfort GCD at discharge were discharged home.
Association between GCD at discharge and readmission in 30 days
Interpretation
In this study, we found that GCD type early during hospitalization was associated with resource utilization during that admission. However, contrary to our hypothesis, patients with medical and comfort GCDs had more health care resource use than patients with resuscitative GCDs. Patients with a medical GCD had longer LOS, higher RIW and increased referral to palliative care, but not flagged intervention use. Similar effects were observed for patients with comfort GCD but were not statistically significant, likely owing to the small number of patients with a comfort GCD. A comfort GCD communicates a desired focus on comfort and relief from symptoms. In keeping with this, the flagged interventions used for patients with a comfort GCD were almost entirely symptom relieving in nature, and 90% of patients with a comfort GCD were referred for a palliative care consult. In regard to these outcomes, the care provided to patients is consistent with the preferences communicated by GCD type.
The increase in LOS and RIW for patients with resuscitative to medical to comfort GCDs may be explained by the fact that patients with a medical GCD, and even more so a comfort GCD, were more ill than those with a resuscitative GCD, and despite the inclusion of variables in the model to adjust for illness severity (Clinical Frailty Scale score, weighted Charlson Comorbidity Index score), residual confounding likely remained.
Our models suggest that GCD type at discharge had little to no association with 30-day readmission (RR estimates range 1.06–1.00); however, we lacked power to detect anything but large differences because very few patients with a comfort GCD were discharged home. In our data, discharge GCD was associated with discharge location, but GCD type does not cause a patient to be discharged to any specific location. Discharge location is an expected effect modifier in the relation between GCD type and 30-day readmission, for example, patients with a comfort GCD discharged to hospice being less likely to be readmitted to hospital than those discharged home because of the patient and caregiver supports available in hospice. Only 7 participants entered hospital with a pre-existing comfort GCD (4.9% of participants with a preadmission GCD), and only 2 of 33 (6.1%) patients discharged with a comfort GCD were readmitted in 30 days (v. 15.5% resuscitative GCD and 21.5% medical GCD). Possibly, many patients with a comfort GCD are having their needs met in the community and are successfully managed without transfer to hospital as per preference communicated by that GCD. Studying inpatients with a comfort GCD likely overestimates the resource use of patients with a comfort GCD compared with those in the community. The association between GCD type, transfers and resource use in other locations such as long-term care and home care is of particularly interest but remains to be explored.
A retrospective study involving 1818 decedents found that patients with “comfort-only” medical orders were less likely to be admitted to ICU or receive life-sustaining treatments than “full-treatment” patients.10 We did not replicate this result; however, the overall ICU admission rate was much higher in the previous study,10 and 41% of patients with comfort-only orders still received discordant intensive care,10 suggesting gaps in communication and processes to ensure patient goals are followed. A first ACCEPT study found 30% agreement between patients’ expressed care preferences and documented orders.13 Another study involving trauma patients found that comfort medical orders were not associated with reduced ICU admission, in part because documentation was not acknowledged in a timely fashion.35 This literature highlights the complex process of communication and shared decision-making in determining medical orders and ensuring a robust transfer of information.
Limitations
This study was designed to recruit older adults with a 50% or greater 6-month mortality risk,17,18 but participants needed to have the capacity and energy to consent and participate. The latter requirements meant that patients in the ICU during the enrolment window were excluded (limited capacity) and overall likely skewed enrolment toward patients who were not critically ill. The 3.4% mortality rate of participants with a resuscitative GCD at enrolment speaks to their relative wellness. Consequently, this study may underestimate the resource use consequences of a resuscitative GCD for patients who die in the following 6 months. Furthermore, with the difficulties in enrolling sicker participants, relatively few patients had a comfort GCD, reducing the power to detect statistically significant differences in outcomes. Had this been a retrospective study of deceased inpatients, the effect of GCD type on resource use may have been quite different. Finally, the models run were adjusted for city but not hospital and unit owing to sample size (too few observations in each stratum). Thus, hospital and unit may be confounding the reported associations.
Selection bias introduced by this study’s inclusion and exclusion criteria limit generalizability, particularly with regard to patients discharged from hospital (or died) in under 48 hours, patients with illnesses (and age) not specified in the inclusion criteria, non–English-speakers and those with cognitive impairment. Sampling occurred in Alberta, Canada, and thus results may not be generalizable to other provinces or countries.
The indicators used to evaluate health care resource use: RIW, LOS, ICU admission, flagged interventions and palliative care referral, are not independent, but examining each measure provided a more complete picture of health resource utilization than any individual metric. Patients with the same LOS could have very different resource use if their admissions ended in death or discharge, because the days leading up to death often feature very high resource use. Resource Intensity Weights should better reflect the difference in resource use. In this study, only 7.4% of patients died, so this is unlikely to have had a large effect on the results.
Conclusion
This study examined health resource utilization in relation to the GCD framework, looking for association between patient GCD type (indicting preference for care) and the care received in hospital. Goals of Care Designation type early in admission was associated with some inpatient health care resource use. For example, patients with a comfort GCD were more likely to receive specialist palliative care referral and flagged interventions that provide symptom relief but are not life-prolonging, which is evidence of goal-concordant care. In the future, a population-based retrospective study involving deceased patients across care sectors (from home to hospital), in which participants can be aligned on time to death, may reduce confounding due to illness severity, and improve estimation of the association between GCD type and health resource use.
Acknowledgements
The authors acknowledge the Advance Care Planning Collaborative Research and Innovation Opportunities network membership, the research assistants, hospital staff and patients that contributed to this study.
Footnotes
Competing interests: Jessica Simon reports grants from Canadian Institutes of Health Research, Alberta Health, Canadian Foundation for Healthcare Improvement and Choosing Wisely Alberta via its partners: the Alberta Medical Association, Alberta Health, Alberta Health Services and the College of Physicians and Surgeons of Alberta. Dr. Simon reports speaker honoraria from Queen’s University and a role as physician consultant, Advance Care Planning and Goals of Care, Alberta Health Services, Calgary Zone. Dr. Simon also reports unpaid roles as co-chair, Advisory Committee, Advance Care Planning Alberta; advisory committee member, Advance Care Planning Canada; and committee member, Advance Care Planning International. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Madalene Earp, Jessica Simon and Konrad Fassbender had full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Konrad Fassbender, Madalene Earp, Jessica Simon and Sara Davison contributed to concept and design. Seema King, Winnie Sia, Amanda Brisebois, LeAnn Esau, Maureen Douglas, Patricia Biondo, Jessica Simon, Konrad Fassbender and Madalene Earp acquired, analyzed or interpreted data. Madalene Earp, Jessica Simon, Konrad Fassbender and Maureen Douglas drafted the manuscript. Madalene Earp performed the statistical analysis. Jessica Simon and Konrad Fassbender obtained funding. Maureen Douglas, Patricia Biondo, LeAnn Esau, Sara Davison, Amanda Brisebois and Winnie Sia provided administrative, technical or material support. Jessica Simon supervised the study. All authors revised the manuscript for intellectual content. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was supported by an Alberta Innovates Collaborative Research and Innovation Opportunities Grant (Program Grant #201201157). The funders had no role in the design, interpretation of results or decision to publish.
Data sharing: Data-sharing requests for deidentified data reported in this article will be considered on written request to the corresponding author for up to 36 months after publication of this work. Data will be available subject to a written proposal, approval by our local ethics review board and a signed data-sharing agreement.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/4/E945/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors